This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
2fmp
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
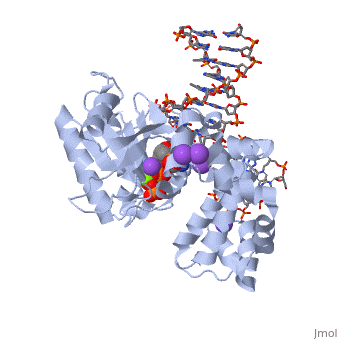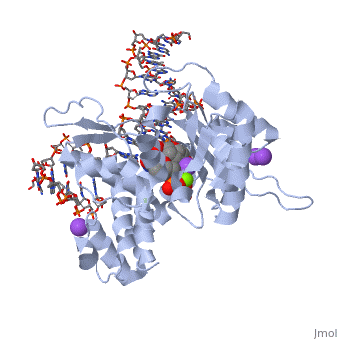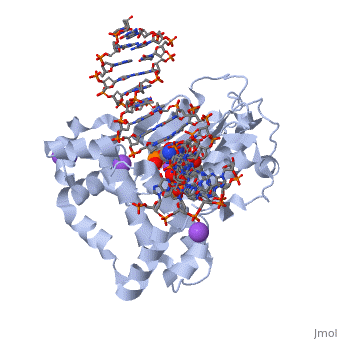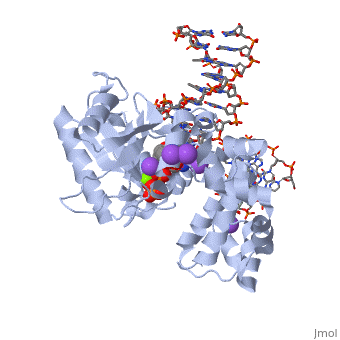
| - | [[ | + | ==DNA Polymerase beta with a terminated gapped DNA substrate and ddCTP with sodium in the catalytic site== |
| + | <StructureSection load='2fmp' size='340' side='right' caption='[[2fmp]], [[Resolution|resolution]] 1.65Å' scene=''> | ||
| + | == Structural highlights == | ||
| + | <table><tr><td colspan='2'>[[2fmp]] is a 4 chain structure with sequence from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2FMP OCA]. For a <b>guided tour on the structure components</b> use [http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=2FMP FirstGlance]. <br> | ||
| + | </td></tr><tr><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=DCT:2,3-DIDEOXYCYTIDINE+5-TRIPHOSPHATE'>DCT</scene>, <scene name='pdbligand=MG:MAGNESIUM+ION'>MG</scene>, <scene name='pdbligand=NA:SODIUM+ION'>NA</scene><br> | ||
| + | <tr><td class="sblockLbl"><b>[[Non-Standard_Residue|NonStd Res:]]</b></td><td class="sblockDat"><scene name='pdbligand=DOC:2,3-DIDEOXYCYTIDINE-5-MONOPHOSPHATE'>DOC</scene></td></tr> | ||
| + | <tr><td class="sblockLbl"><b>[[Related_structure|Related:]]</b></td><td class="sblockDat">[[2fmq|2fmq]], [[2fms|2fms]]</td></tr> | ||
| + | <tr><td class="sblockLbl"><b>[[Gene|Gene:]]</b></td><td class="sblockDat">POLB ([http://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Info&srchmode=5&id=9606 Homo sapiens])</td></tr> | ||
| + | <tr><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=2fmp FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2fmp OCA], [http://www.rcsb.org/pdb/explore.do?structureId=2fmp RCSB], [http://www.ebi.ac.uk/pdbsum/2fmp PDBsum]</span></td></tr> | ||
| + | <table> | ||
| + | == Evolutionary Conservation == | ||
| + | [[Image:Consurf_key_small.gif|200px|right]] | ||
| + | Check<jmol> | ||
| + | <jmolCheckbox> | ||
| + | <scriptWhenChecked>select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/fm/2fmp_consurf.spt"</scriptWhenChecked> | ||
| + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
| + | <text>to colour the structure by Evolutionary Conservation</text> | ||
| + | </jmolCheckbox> | ||
| + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/chain_selection.php?pdb_ID=2ata ConSurf]. | ||
| + | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
| + | The molecular details of the nucleotidyl transferase reaction have remained speculative, as strategies to trap catalytic intermediates for structure determination utilize substrates lacking the primer terminus 3'-OH and catalytic Mg2+, resulting in an incomplete and distorted active site geometry. Since the geometric arrangement of these essential atoms will impact chemistry, structural insight into fidelity strategies has been hampered. Here, we present a crystal structure of a precatalytic complex of a DNA polymerase with bound substrates that include the primer 3'-OH and catalytic Mg2+. This catalytic intermediate was trapped with a nonhydrolyzable deoxynucleotide analog. Comparison with two new structures of DNA polymerase beta lacking the 3'-OH or catalytic Mg2+ is described. These structures provide direct evidence that both atoms are required to achieve a proper geometry necessary for an in-line nucleophilic attack of O3' on the alphaP of the incoming nucleotide. | ||
| - | + | Magnesium-induced assembly of a complete DNA polymerase catalytic complex.,Batra VK, Beard WA, Shock DD, Krahn JM, Pedersen LC, Wilson SH Structure. 2006 Apr;14(4):757-66. PMID:16615916<ref>PMID:16615916</ref> | |
| - | + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |
| - | + | </div> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
==See Also== | ==See Also== | ||
*[[DNA polymerase|DNA polymerase]] | *[[DNA polymerase|DNA polymerase]] | ||
| - | + | == References == | |
| - | == | + | <references/> |
| - | < | + | __TOC__ |
| + | </StructureSection> | ||
[[Category: Homo sapiens]] | [[Category: Homo sapiens]] | ||
[[Category: Batra, V K.]] | [[Category: Batra, V K.]] | ||
Revision as of 04:34, 29 September 2014
DNA Polymerase beta with a terminated gapped DNA substrate and ddCTP with sodium in the catalytic site
| |||||||||||