This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
3i2t
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
| - | [[ | + | ==Crystal structure of the unliganded Drosophila Epidermal Growth Factor Receptor ectodomain== |
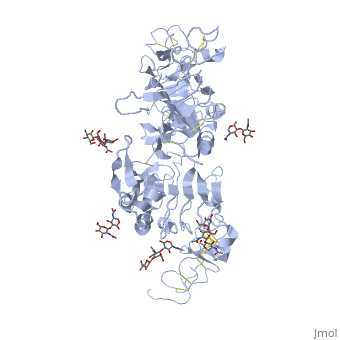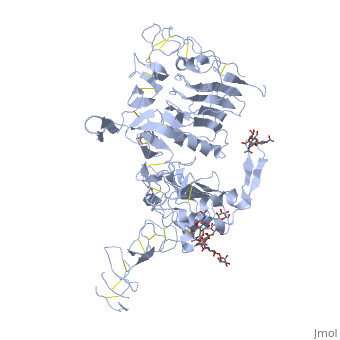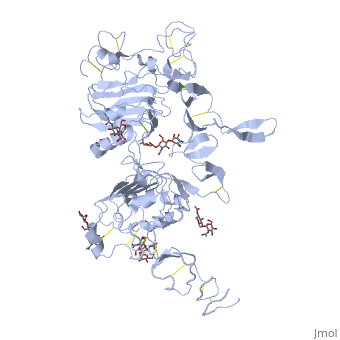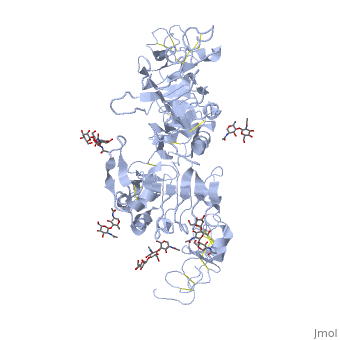
| + | <StructureSection load='3i2t' size='340' side='right' caption='[[3i2t]], [[Resolution|resolution]] 2.70Å' scene=''> | ||
| + | == Structural highlights == | ||
| + | <table><tr><td colspan='2'>[[3i2t]] is a 1 chain structure with sequence from [http://en.wikipedia.org/wiki/Drosophila_melanogaster Drosophila melanogaster]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=3I2T OCA]. For a <b>guided tour on the structure components</b> use [http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=3I2T FirstGlance]. <br> | ||
| + | </td></tr><tr><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=BMA:BETA-D-MANNOSE'>BMA</scene>, <scene name='pdbligand=NAG:N-ACETYL-D-GLUCOSAMINE'>NAG</scene>, <scene name='pdbligand=NDG:2-(ACETYLAMINO)-2-DEOXY-A-D-GLUCOPYRANOSE'>NDG</scene><br> | ||
| + | <tr><td class="sblockLbl"><b>[[Gene|Gene:]]</b></td><td class="sblockDat">Egfr ([http://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Info&srchmode=5&id=7227 Drosophila melanogaster])</td></tr> | ||
| + | <tr><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=3i2t FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=3i2t OCA], [http://www.rcsb.org/pdb/explore.do?structureId=3i2t RCSB], [http://www.ebi.ac.uk/pdbsum/3i2t PDBsum]</span></td></tr> | ||
| + | <table> | ||
| + | == Evolutionary Conservation == | ||
| + | [[Image:Consurf_key_small.gif|200px|right]] | ||
| + | Check<jmol> | ||
| + | <jmolCheckbox> | ||
| + | <scriptWhenChecked>select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/i2/3i2t_consurf.spt"</scriptWhenChecked> | ||
| + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
| + | <text>to colour the structure by Evolutionary Conservation</text> | ||
| + | </jmolCheckbox> | ||
| + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/chain_selection.php?pdb_ID=2ata ConSurf]. | ||
| + | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
| + | The orphan receptor tyrosine kinase ErbB2 (also known as HER2 or Neu) transforms cells when overexpressed, and it is an important therapeutic target in human cancer. Structural studies have suggested that the oncogenic (and ligand-independent) signalling properties of ErbB2 result from the absence of a key intramolecular 'tether' in the extracellular region that autoinhibits other human ErbB receptors, including the epidermal growth factor (EGF) receptor. Although ErbB2 is unique among the four human ErbB receptors, here we show that it is the closest structural relative of the single EGF receptor family member in Drosophila melanogaster (dEGFR). Genetic and biochemical data show that dEGFR is tightly regulated by growth factor ligands, yet a crystal structure shows that it, too, lacks the intramolecular tether seen in human EGFR, ErbB3 and ErbB4. Instead, a distinct set of autoinhibitory interdomain interactions hold unliganded dEGFR in an inactive state. All of these interactions are maintained (and even extended) in ErbB2, arguing against the suggestion that ErbB2 lacks autoinhibition. We therefore suggest that normal and pathogenic ErbB2 signalling may be regulated by ligands in the same way as dEGFR. Our findings have important implications for ErbB2 regulation in human cancer, and for developing therapeutic approaches that target novel aspects of this orphan receptor. | ||
| - | + | ErbB2 resembles an autoinhibited invertebrate epidermal growth factor receptor.,Alvarado D, Klein DE, Lemmon MA Nature. 2009 Sep 10;461(7261):287-91. Epub 2009 Aug 30. PMID:19718021<ref>PMID:19718021</ref> | |
| - | + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |
| - | + | </div> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
==See Also== | ==See Also== | ||
*[[Epidermal Growth Factor Receptor|Epidermal Growth Factor Receptor]] | *[[Epidermal Growth Factor Receptor|Epidermal Growth Factor Receptor]] | ||
| - | + | == References == | |
| - | == | + | <references/> |
| - | < | + | __TOC__ |
| + | </StructureSection> | ||
[[Category: Drosophila melanogaster]] | [[Category: Drosophila melanogaster]] | ||
[[Category: Alvarado, D.]] | [[Category: Alvarado, D.]] | ||
Revision as of 13:05, 29 September 2014
Crystal structure of the unliganded Drosophila Epidermal Growth Factor Receptor ectodomain
| |||||||||||