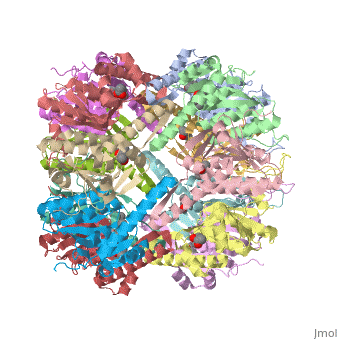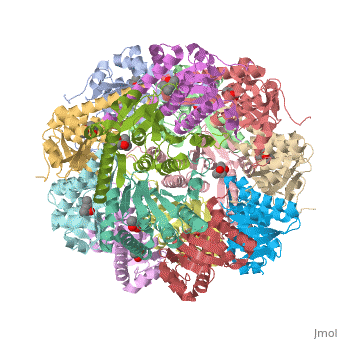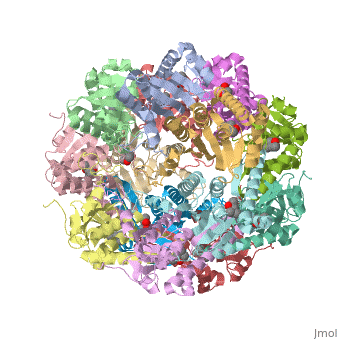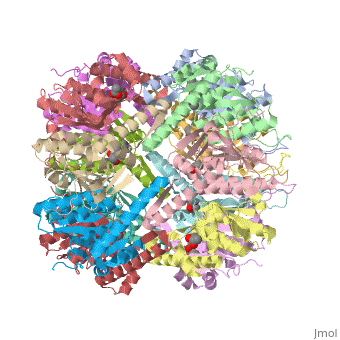
Molecular Playground/E. coli ClpP
From Proteopedia
(Difference between revisions)
| Line 12: | Line 12: | ||
This is a sample scene created with SAT to <scene name="/12/3456/Sample/1">color</scene> by Group, and another to make <scene name="/12/3456/Sample/2">a transparent representation</scene> of the protein. You can make your own scenes on SAT starting from scratch or loading and editing one of these sample scenes. | This is a sample scene created with SAT to <scene name="/12/3456/Sample/1">color</scene> by Group, and another to make <scene name="/12/3456/Sample/2">a transparent representation</scene> of the protein. You can make your own scenes on SAT starting from scratch or loading and editing one of these sample scenes. | ||
== Role of ClpP/Biological Relevance == | == Role of ClpP/Biological Relevance == | ||
| - | ''E. coli'' ClpAP/ClpXP complexes play a critical role in maintaining protein homeostasis under several levels of quality control. Improperly folded or aggregated proteins are potential ClpP substrates based on properties of the associated regulatory element recognition. Targeted removal of aberrant proteins resulting from and rescue of stalled ribosomes by the SsrA tagging system are directly recognized and degraded by ClpAP/ClpXP complexes [9]. In E. coli ClpP and ClpP homologues found in other bacteria require regulatory elements to recognize and import proteins for destruction. To gain access to the active sites is tightly controlled and therefore a potential antimicrobial target where loss of regulation (for example, through use of acyldepsipeptides or ADEPs) literally digests the bacteria from the inside out [10]. E. coli ClpAP and ClpXP has been used as a structural model for the 26 proteasome to gain insight into the workings [11,12]. | + | ''E. coli'' ClpAP/ClpXP complexes play a critical role in maintaining protein homeostasis under several levels of quality control. Improperly folded or aggregated proteins are potential ClpP substrates based on properties of the associated regulatory element recognition. Targeted removal of aberrant proteins resulting from and rescue of stalled ribosomes by the SsrA tagging system are directly recognized and degraded by ClpAP/ClpXP complexes [9]. In ''E. coli'' ClpP and ClpP homologues found in other bacteria require regulatory elements to recognize and import proteins for destruction. To gain access to the active sites is tightly controlled and therefore a potential antimicrobial target where loss of regulation (for example, through use of acyldepsipeptides or ADEPs) literally digests the bacteria from the inside out [10]. ''E. coli'' ClpAP and ClpXP has been used as a structural model for the 26 proteasome to gain insight into the workings [11,12]. |
</StructureSection> | </StructureSection> | ||
Revision as of 19:48, 3 December 2014
Here in the Chien lab, we study how proteolysis plays a large part in protein quality control. The maintenance and timely destruction of protein levels plays an important role during cell homeostasis and cell transitions/differentiation, yet much of what governs these processes has yet to be fully understood.
| |||||||||||
References
Acknowledgements
Kamal Joshi, Joanne Lau, Jing Liu, Rob Vass