This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1pgo
From Proteopedia
| Line 1: | Line 1: | ||
| - | [[Image:1pgo.jpg|left|200px]] | + | [[Image:1pgo.jpg|left|200px]] |
| - | + | ||
| - | '''CRYSTALLOGRAPHIC STUDY OF COENZYME, COENZYME ANALOGUE AND SUBSTRATE BINDING IN 6-PHOSPHOGLUCONATE DEHYDROGENASE: IMPLICATIONS FOR NADP SPECIFICITY AND THE ENZYME MECHANISM''' | + | {{Structure |
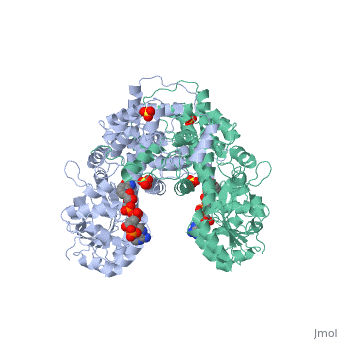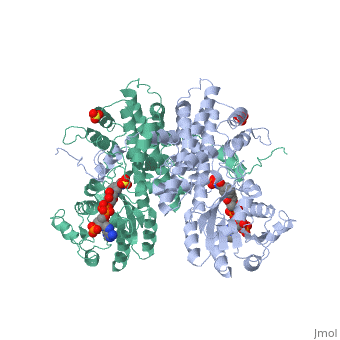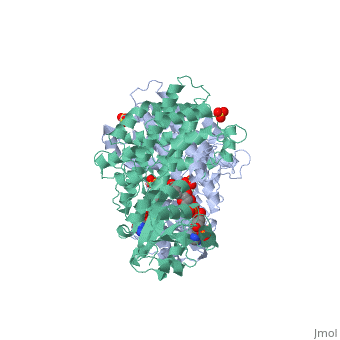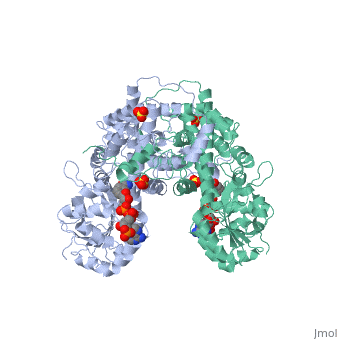
| + | |PDB= 1pgo |SIZE=350|CAPTION= <scene name='initialview01'>1pgo</scene>, resolution 2.5Å | ||
| + | |SITE= | ||
| + | |LIGAND= <scene name='pdbligand=SO4:SULFATE+ION'>SO4</scene> and <scene name='pdbligand=NDP:NADPH DIHYDRO-NICOTINAMIDE-ADENINE-DINUCLEOTIDE PHOSPHATE'>NDP</scene> | ||
| + | |ACTIVITY= [http://en.wikipedia.org/wiki/Phosphogluconate_dehydrogenase_(decarboxylating) Phosphogluconate dehydrogenase (decarboxylating)], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=1.1.1.44 1.1.1.44] | ||
| + | |GENE= | ||
| + | }} | ||
| + | |||
| + | '''CRYSTALLOGRAPHIC STUDY OF COENZYME, COENZYME ANALOGUE AND SUBSTRATE BINDING IN 6-PHOSPHOGLUCONATE DEHYDROGENASE: IMPLICATIONS FOR NADP SPECIFICITY AND THE ENZYME MECHANISM''' | ||
| + | |||
==Overview== | ==Overview== | ||
| Line 7: | Line 16: | ||
==About this Structure== | ==About this Structure== | ||
| - | 1PGO is a [ | + | 1PGO is a [[Single protein]] structure of sequence from [http://en.wikipedia.org/wiki/Ovis_aries Ovis aries]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1PGO OCA]. |
==Reference== | ==Reference== | ||
| - | Crystallographic study of coenzyme, coenzyme analogue and substrate binding in 6-phosphogluconate dehydrogenase: implications for NADP specificity and the enzyme mechanism., Adams MJ, Ellis GH, Gover S, Naylor CE, Phillips C, Structure. 1994 Jul 15;2(7):651-68. PMID:[http:// | + | Crystallographic study of coenzyme, coenzyme analogue and substrate binding in 6-phosphogluconate dehydrogenase: implications for NADP specificity and the enzyme mechanism., Adams MJ, Ellis GH, Gover S, Naylor CE, Phillips C, Structure. 1994 Jul 15;2(7):651-68. PMID:[http://www.ncbi.nlm.nih.gov/pubmed/7922042 7922042] |
[[Category: Ovis aries]] | [[Category: Ovis aries]] | ||
[[Category: Phosphogluconate dehydrogenase (decarboxylating)]] | [[Category: Phosphogluconate dehydrogenase (decarboxylating)]] | ||
| Line 21: | Line 30: | ||
[[Category: oxidoreductase (choh(d)-nadp+(a))]] | [[Category: oxidoreductase (choh(d)-nadp+(a))]] | ||
| - | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Mar 20 13:24:06 2008'' |
Revision as of 11:24, 20 March 2008
| |||||||
| , resolution 2.5Å | |||||||
|---|---|---|---|---|---|---|---|
| Ligands: | and | ||||||
| Activity: | Phosphogluconate dehydrogenase (decarboxylating), with EC number 1.1.1.44 | ||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||
CRYSTALLOGRAPHIC STUDY OF COENZYME, COENZYME ANALOGUE AND SUBSTRATE BINDING IN 6-PHOSPHOGLUCONATE DEHYDROGENASE: IMPLICATIONS FOR NADP SPECIFICITY AND THE ENZYME MECHANISM
Overview
BACKGROUND: The nicotinamide adenine dinucleotide phosphate (NADP)-dependent oxidative decarboxylase, 6-phosphogluconate dehydrogenase, is a major source of reduced coenzyme for synthesis. Enzymes later in the pentose phosphate pathway convert the reaction product, ribulose 5-phosphate, to ribose 5-phosphate. Crystallographic study of complexes with coenzyme and substrate explain the NADP dependence which determines the enzyme's metabolic role and support the proposed general base-general acid mechanism. RESULTS: The refined structures of binary coenzyme/analogue complexes show that Arg33 is ordered by binding the 2'-phosphate, and provides one face of the adenine site. The nicotinamide, while less tightly bound, is more extended when reduced than when oxidized. All substrate binding residues are conserved; the 3-hydroxyl of 6-phosphogluconate is hydrogen bonded to N zeta of Lys183 and the 3-hydrogen points towards the oxidized nicotinamide. The 6-phosphate replaces a tightly bound sulphate in the apo-enzyme. CONCLUSIONS: NADP specificity is achieved primarily by Arg33 which binds the 2'-phosphate but, in its absence, obscures the adenine pocket. The bound oxidized nicotinamide is syn; hydride transfer from bound substrate to the nicotinamide si- face is achieved with a small movement of the nicotinamide nucleotide. Lys183 may act as general base. A water bound to Gly130 in the coenzyme domain is the most likely acid required in decarboxylation. The dihydronicotinamide ring of NADPH competes for ligands with the 1-carboxyl of 6-phosphogluconate.
About this Structure
1PGO is a Single protein structure of sequence from Ovis aries. Full crystallographic information is available from OCA.
Reference
Crystallographic study of coenzyme, coenzyme analogue and substrate binding in 6-phosphogluconate dehydrogenase: implications for NADP specificity and the enzyme mechanism., Adams MJ, Ellis GH, Gover S, Naylor CE, Phillips C, Structure. 1994 Jul 15;2(7):651-68. PMID:7922042
Page seeded by OCA on Thu Mar 20 13:24:06 2008