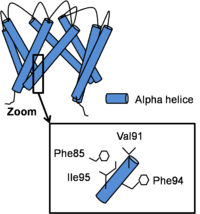This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 969
From Proteopedia
(Difference between revisions)
| Line 19: | Line 19: | ||
In response to a '''external stimuli''', the structure of the NaK channel is different. In fact, after some inter- and intra-subunit rearrangements, the NaK channel can be '''open or closed'''. | In response to a '''external stimuli''', the structure of the NaK channel is different. In fact, after some inter- and intra-subunit rearrangements, the NaK channel can be '''open or closed'''. | ||
| + | [[Image:closed_structure.png|right|thumbnail|200px|'''The structure of the channel in his closed conformation''']] | ||
====Closed Conformation==== | ====Closed Conformation==== | ||
In the closed conformation, inner helices are near and straight. There is a subsequent '''bundle crossing''' formed by interactions between C-terminal residues. In the region just above the bundle crossing, <scene name='60/604488/Phe_92/1'>Phe92</scene> from each inner helix forms contacts with a '''hydrophobic patch''' on the opposite face of Phe92 from the neighboring inner helix formed by Val91, Phe94, Ile95 and Leu98. <ref> PMID: 19098917</ref> | In the closed conformation, inner helices are near and straight. There is a subsequent '''bundle crossing''' formed by interactions between C-terminal residues. In the region just above the bundle crossing, <scene name='60/604488/Phe_92/1'>Phe92</scene> from each inner helix forms contacts with a '''hydrophobic patch''' on the opposite face of Phe92 from the neighboring inner helix formed by Val91, Phe94, Ile95 and Leu98. <ref> PMID: 19098917</ref> | ||
| - | [[Image:closed_structure.jpg|right|thumbnail|300px|'''The structure of the channel in his closed conformation''']] | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | [[Image:open_structure.png|right|thumbnail|200px|'''The structure of the channel in his open conformation''']] | ||
====Open Conformation==== | ====Open Conformation==== | ||
Channel opening is a '''conserved mechanism'''.The inner helix '''twist''' and '''bend''' thanks to a<scene name='60/604488/Glycine_conserved/1'> conserved glycine residue Gly87</scene> which is considered as the '''gating hinge'''. After this bending, the inner helices twist of 45° around their helical helix and the outer helix tilt tangentially in the same direction by 11° without any twisting motion. As all of helix twist or move inside of a subunit, intra-subunit interactions between inner and outer helix don’t differ a lot. On the contrary, inter-subunit interactions between neighboring inner helix change. In fact, Phe92 swings away and points its side chain towards the central ion conduction pathway due to inner helix bending and the hydrophobic patch slides along the neighboring inner helix by two helical turns and forms new Van der Waals contacts with <scene name='60/604488/Phe85/1'>Phe 85 </scene>. This resulted in a '''disruption of the bundle crossing''' and so intra- and inter- subunits interactions in the open state become less important than in the close state. <ref>PMID: 19098917</ref> | Channel opening is a '''conserved mechanism'''.The inner helix '''twist''' and '''bend''' thanks to a<scene name='60/604488/Glycine_conserved/1'> conserved glycine residue Gly87</scene> which is considered as the '''gating hinge'''. After this bending, the inner helices twist of 45° around their helical helix and the outer helix tilt tangentially in the same direction by 11° without any twisting motion. As all of helix twist or move inside of a subunit, intra-subunit interactions between inner and outer helix don’t differ a lot. On the contrary, inter-subunit interactions between neighboring inner helix change. In fact, Phe92 swings away and points its side chain towards the central ion conduction pathway due to inner helix bending and the hydrophobic patch slides along the neighboring inner helix by two helical turns and forms new Van der Waals contacts with <scene name='60/604488/Phe85/1'>Phe 85 </scene>. This resulted in a '''disruption of the bundle crossing''' and so intra- and inter- subunits interactions in the open state become less important than in the close state. <ref>PMID: 19098917</ref> | ||
| - | + | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
== '''Active Site & Ions Passage''' == | == '''Active Site & Ions Passage''' == | ||
Revision as of 17:24, 8 January 2015
NaK channel
| |||||||||||