This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 951
From Proteopedia
(Difference between revisions)
| Line 15: | Line 15: | ||
== Global Structure == | == Global Structure == | ||
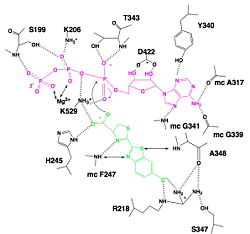
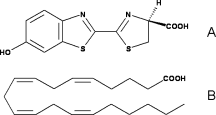
| - | Luciferase is a 62kDa protein. It contains 550 amino acids. The enzyme can be divide into two domains. On the one hand, the major portion, corresponding to the <scene name='60/604470/N_terminal_domain/1'>N-terminal</scene>. On the other hand, the small portion, corresponding to the <scene name='60/604470/C_terminal_domain/1'>C-terminal domain</scene>.Those two domains are separated by a large cleft.<ref>PMID:8805533</ref> | + | Luciferase is a 62kDa protein. It contains 550 amino acids. The enzyme can be divide into two domains. On the one hand, the major portion, corresponding to the <scene name='60/604470/N_terminal_domain/1'>N-terminal</scene>. On the other hand, the small portion, corresponding to the <scene name='60/604470/C_terminal_domain/1'>C-terminal domain</scene>.Those two domains are separated by a large cleft.<ref name =''first''>PMID:8805533</ref> |
====The C terminal domain==== | ====The C terminal domain==== | ||
| - | The amino acids of C terminal sequence is composed of contiguous residues and form a type of lid upon the N terminal domain. It contains two β sheets: the <scene name='60/604470/Beta_sheet/2'>first β sheet </scene> is composed of two short antiparallel strands and the <scene name='60/604470/Beta_sheet_2/1'> second β sheet</scene> is composed of 3 antiparallel strands, mixed with <scene name='60/604470/3_alpha_helices/1'>three α helices</scene>. Those helices are put toward the outside.It is α + β structure.<ref>PMID:8805533</ref> | + | The amino acids of C terminal sequence is composed of contiguous residues and form a type of lid upon the N terminal domain. It contains two β sheets: the <scene name='60/604470/Beta_sheet/2'>first β sheet </scene> is composed of two short antiparallel strands and the <scene name='60/604470/Beta_sheet_2/1'> second β sheet</scene> is composed of 3 antiparallel strands, mixed with <scene name='60/604470/3_alpha_helices/1'>three α helices</scene>. Those helices are put toward the outside.It is α + β structure.<ref name =''second''>PMID:8805533</ref> |
====The N terminal domain==== | ====The N terminal domain==== | ||
| - | The amino acids of C terminal sequence is composed of non contiguous residues. It falls into three subdomains (<scene name='60/604470/Domain_a/1'>the subdomain A</scene>,<scene name='60/604470/Domain_b/1'>the subdomain B</scene>, and <scene name='60/604470/Domain_c/1'>the subdomain C</scene>).It has an antiparallel <scene name='60/604470/Beta_barrel/3'>β barrel</scene>, two β sheets, which are framed by <scene name='60/604470/Alpha_helices/1'>α helices</scene>. Each of the two β sheets subdomains (A and B)are composed of 8 β strands and 6 helices. The β sheet A has <scene name='60/604470/A_beta_strands/1'>5 parallel and 3 antiparallel β strands</scene>. The β sheet B has <scene name='60/604470/A_beta_strands/1'>6 parallel and 2 antiparallel β strands</scene>. Those β sheets create a groove, closed on one end by the β barrel.<ref>PMID:8805533</ref> | + | The amino acids of C terminal sequence is composed of non contiguous residues. It falls into three subdomains (<scene name='60/604470/Domain_a/1'>the subdomain A</scene>,<scene name='60/604470/Domain_b/1'>the subdomain B</scene>, and <scene name='60/604470/Domain_c/1'>the subdomain C</scene>).It has an antiparallel <scene name='60/604470/Beta_barrel/3'>β barrel</scene>, two β sheets, which are framed by <scene name='60/604470/Alpha_helices/1'>α helices</scene>. Each of the two β sheets subdomains (A and B)are composed of 8 β strands and 6 helices. The β sheet A has <scene name='60/604470/A_beta_strands/1'>5 parallel and 3 antiparallel β strands</scene>. The β sheet B has <scene name='60/604470/A_beta_strands/1'>6 parallel and 2 antiparallel β strands</scene>. Those β sheets create a groove, closed on one end by the β barrel.<ref name =''third''>PMID:8805533</ref> |
Revision as of 11:00, 9 January 2015
| |||||||||||
References
- ↑ Marques SM, Esteves da Silva JC. Firefly bioluminescence: a mechanistic approach of luciferase catalyzed reactions. IUBMB Life. 2009 Jan;61(1):6-17. PMID:18949818 doi:10.1002/iub.134
- ↑ 2.0 2.1 2.2 Conti E, Franks NP, Brick P. Crystal structure of firefly luciferase throws light on a superfamily of adenylate-forming enzymes. Structure. 1996 Mar 15;4(3):287-98. PMID:8805533
- ↑ Conti E, Franks NP, Brick P. Crystal structure of firefly luciferase throws light on a superfamily of adenylate-forming enzymes. Structure. 1996 Mar 15;4(3):287-98. PMID:8805533
- ↑ Photobiology
- ↑ Conti E, Franks NP, Brick P. Crystal structure of firefly luciferase throws light on a superfamily of adenylate-forming enzymes. Structure. 1996 Mar 15;4(3):287-98. PMID:8805533
- ↑ Photobiology
- ↑ Conti E, Franks NP, Brick P. Crystal structure of firefly luciferase throws light on a superfamily of adenylate-forming enzymes. Structure. 1996 Mar 15;4(3):287-98. PMID:8805533
- ↑ Photobiology
- ↑ Marques SM, Esteves da Silva JC. Firefly bioluminescence: a mechanistic approach of luciferase catalyzed reactions. IUBMB Life. 2009 Jan;61(1):6-17. PMID:18949818 doi:10.1002/iub.134
- ↑ Hosseinkhani S. Molecular enigma of multicolor bioluminescence of firefly luciferase. Cell Mol Life Sci. 2011 Apr;68(7):1167-82. doi: 10.1007/s00018-010-0607-0. Epub, 2010 Dec 28. PMID:21188462 doi:http://dx.doi.org/10.1007/s00018-010-0607-0
- ↑ Inouye S. Firefly luciferase: an adenylate-forming enzyme for multicatalytic functions. Cell Mol Life Sci. 2010 Feb;67(3):387-404. Epub 2009 Oct 27. PMID:19859663 doi:10.1007/s00018-009-0170-8