This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 972
From Proteopedia
(Difference between revisions)
| Line 38: | Line 38: | ||
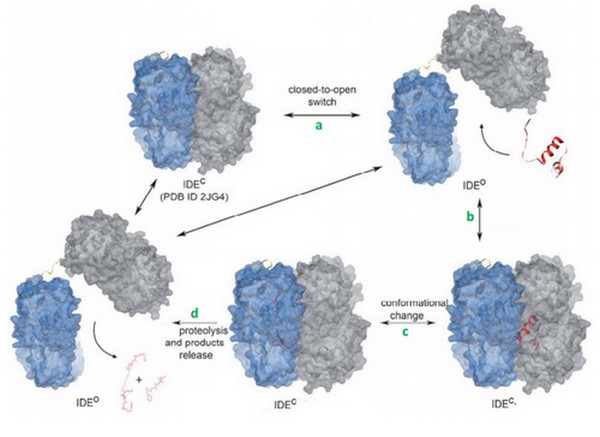
IDE catalytic site has a high affinity for hydrophobic and basic. Bradykinin is essentially composed by proline and arginine, which are basic amino acids. So, bradykinin structure may explain this strange interaction. | IDE catalytic site has a high affinity for hydrophobic and basic. Bradykinin is essentially composed by proline and arginine, which are basic amino acids. So, bradykinin structure may explain this strange interaction. | ||
| - | |||
| - | |||
Crystal structure revealed that <scene name='60/604491/Residues_336_to_342/1'>residues 336 to 342</scene> and <scene name='60/604491/Residues_359_to_369/1'>residues 359 to 369</scene> of IDE are involved in interactions with bradykinin. | Crystal structure revealed that <scene name='60/604491/Residues_336_to_342/1'>residues 336 to 342</scene> and <scene name='60/604491/Residues_359_to_369/1'>residues 359 to 369</scene> of IDE are involved in interactions with bradykinin. | ||
N-ter 3 residues of bradykinin (Arg1, Pro2, Pro3) is also found to interact with the exosite. | N-ter 3 residues of bradykinin (Arg1, Pro2, Pro3) is also found to interact with the exosite. | ||
| + | |||
| + | Im et al<ref>doi: 10.1074/jbc.M701590200</ref> suggested that ATP increases the activtity of IDE with its small substrates like braydkinin. | ||
==Hypothetical role of bradykinin on IDE== | ==Hypothetical role of bradykinin on IDE== | ||
Revision as of 21:31, 9 January 2015
| This Sandbox is Reserved from 15/11/2014, through 15/05/2015 for use in the course "Biomolecule" taught by Bruno Kieffer at the Strasbourg University. This reservation includes Sandbox Reserved 951 through Sandbox Reserved 975. |
To get started:
More help: Help:Editing |
| |||||||||||
References
- ↑ Song ES, Juliano MA, Juliano L, Hersh LB. Substrate activation of insulin-degrading enzyme (insulysin). A potential target for drug development. J Biol Chem. 2003 Dec 12;278(50):49789-94. Epub 2003 Oct 2. PMID:14527953 doi:http://dx.doi.org/10.1074/jbc.M308983200
- ↑ Im H, Manolopoulou M, Malito E, Shen Y, Zhao J, Neant-Fery M, Sun CY, Meredith SC, Sisodia SS, Leissring MA, Tang WJ. Structure of substrate-free human insulin-degrading enzyme (IDE) and biophysical analysis of ATP-induced conformational switch of IDE. J Biol Chem. 2007 Aug 31;282(35):25453-63. Epub 2007 Jul 5. PMID:17613531 doi:10.1074/jbc.M701590200
- ↑ Malito E, Ralat LA, Manolopoulou M, Tsay JL, Wadlington NL, Tang WJ. Molecular Bases for the Recognition of Short Peptide Substrates and Cysteine-Directed Modifications of Human Insulin-Degrading Enzyme. Biochemistry. 2008 Nov 6. PMID:18986166 doi:10.1021/bi801192h