Odorant binding protein
From Proteopedia
| Line 43: | Line 43: | ||
====Protein conformations==== | ====Protein conformations==== | ||
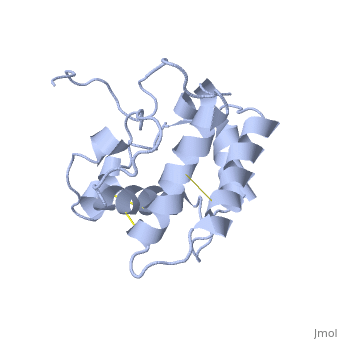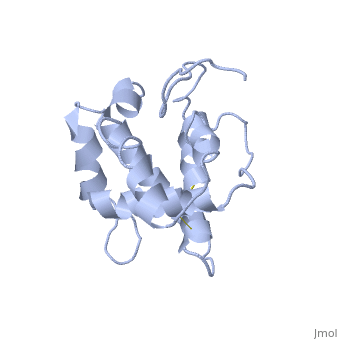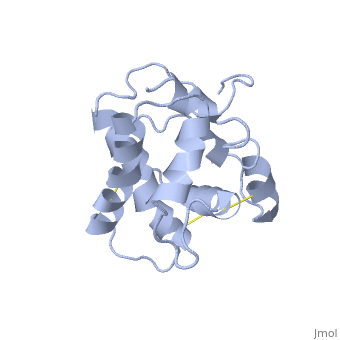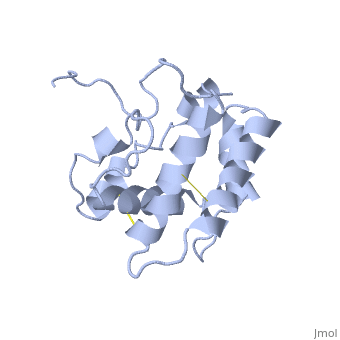
| - | [[Image:A -B forms without ligand.png|thumb|upright=1|The A and B forms without ligand (PDB IDs: [ | + | [[Image:A -B forms without ligand.png|thumb|upright=1|The A and B forms without ligand (PDB IDs: [[1gm0]] and [[1ls8]]).]] |
BmorPBP has two conformations: The "open form" (A) and the "close form" (B)<ref>DOI: 10.1074/jbc.274.43.30950</ref>. The bombykol and the alpha-helix loacated in the c-terminus of the protein compete for the binding site: when the c-terminus is inside the binding cavity it get's an alpha helix shape, and the protien is in its "close form" (B), wherease in the "open form" (A) the c-terminus is outside of the protein and has no defined secondery sturcture. Binding experiments have shown that the B-form binds 15 times higher than the A-form <ref>doi: 10.1073/pnas.0501447102</ref>, therefore considered to be the carrier of the pheromone. The complex of the A-form and the pheromone, is then considered the form that activates the receptor. | BmorPBP has two conformations: The "open form" (A) and the "close form" (B)<ref>DOI: 10.1074/jbc.274.43.30950</ref>. The bombykol and the alpha-helix loacated in the c-terminus of the protein compete for the binding site: when the c-terminus is inside the binding cavity it get's an alpha helix shape, and the protien is in its "close form" (B), wherease in the "open form" (A) the c-terminus is outside of the protein and has no defined secondery sturcture. Binding experiments have shown that the B-form binds 15 times higher than the A-form <ref>doi: 10.1073/pnas.0501447102</ref>, therefore considered to be the carrier of the pheromone. The complex of the A-form and the pheromone, is then considered the form that activates the receptor. | ||
| - | In short, the B-form (c-terminus outside the cavity) occurse only at neutral pH and in the presence of the ligand. The A-form (c-terminus inside the cavity) occurs at both low and nuetral pH, yet at the latter only in the absence of ligand. | + | In short, the B-form (c-terminus outside the cavity) occurse only at neutral pH and in the presence of the ligand. The A-form (c-terminus inside the cavity) occurs at both low and nuetral pH, yet at the latter only in the absence of ligand. Therfor, in neutarl pH when the ligand is binding to the protein in its A-form, the complex formation causes a change in conforamtion to the B-form. |
The transition between the two conformation is taking place between 6-5 pH. | The transition between the two conformation is taking place between 6-5 pH. | ||
| - | + | '''Conformation transition mechanism:''' | |
The c-terminus of the protein bears mostly [[nonpolar amino acids]]. Yet on the surface of the helix there are three exceptional amino acids: Asp-132, Glu-137, and Glu-141, which are conserved in moth PBP <ref>doi: 10.1016/j.bbrc.2005.07.176</ref>. Of these, residues [[Asp-132 and Glu-141]] triggers the formation of the α-helix upon protonation at low pH. This what is causing the ejacullate of the ligand from the binding pocket, which is replaced by the formated alpha helix<ref>doi: 10.1016/j.bbrc</ref>. | The c-terminus of the protein bears mostly [[nonpolar amino acids]]. Yet on the surface of the helix there are three exceptional amino acids: Asp-132, Glu-137, and Glu-141, which are conserved in moth PBP <ref>doi: 10.1016/j.bbrc.2005.07.176</ref>. Of these, residues [[Asp-132 and Glu-141]] triggers the formation of the α-helix upon protonation at low pH. This what is causing the ejacullate of the ligand from the binding pocket, which is replaced by the formated alpha helix<ref>doi: 10.1016/j.bbrc</ref>. | ||
| Line 66: | Line 66: | ||
Two theories have been propsed for the activation of the odorant receptors located on the dendrtirte membrane. One theory suggests that the pheromone-PBP complex is needed for the receptor activation, while the second theory argue that the pheromone itself is sufficient for the activation of the receptor. | Two theories have been propsed for the activation of the odorant receptors located on the dendrtirte membrane. One theory suggests that the pheromone-PBP complex is needed for the receptor activation, while the second theory argue that the pheromone itself is sufficient for the activation of the receptor. | ||
| - | [[Image: | + | [[Image:N model extended.png|thumb|upright=1.5|The events prior the neuron excitation, following the "N model" suggested by Kaissling (2009)<ref name="Kaissling 2009" />]] |
*'''Activation by the pheromone alone''' | *'''Activation by the pheromone alone''' | ||
| - | This model is supported by the pH dependent conforamtion transition, that is described above. | + | This model is supported by the pH dependent conforamtion transition, that is described above. The bulk of the sensillar lymph is in nuetral pH (6.5-7), while environment near the dendrite membrane bears a low pH (4.5), due to the negative charges on the surface of the membrane <ref>DOI: 10.1016/0040-8166(84)90004-1</ref>, which cause the accumulation of positively charged kations near the membrane surface (20-50 nm)<ref name="Kaissling 2009" />. According to this model (illustrated in [[figure 1]]), the pheromone is entering the sensillar lymph through a pore in the cuticle, then it can be either degraded by odorant degrading enzymes (ODE) or bind to a PBP (of both forms). Once the complex is arriving to the low pH environment near the dendrite membrane the PBP will shift to the A-form, thereby ejaculating the ligand from the binding pocket, allowing it to activate the OR:CO-OR complex and the cellular signal transduction begins. |
| + | |||
*'''Activation by the complex pheromone-PBP''' | *'''Activation by the complex pheromone-PBP''' | ||
| - | An alternative mode action was proposed for the receptor activation in the drosophila | + | An alternative mode of action was proposed for the receptor activation in the drosophila, where it was found that the complex of pheromone-PBP is required for the activity of pheromone-sensitive neurons <ref>DOI: 10.1016/j.neuron.2004.12.031<ref/><ref>doi: 10.1016/j.cell.2008.04.046<ref/> |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
</StructureSection> | </StructureSection> | ||
Revision as of 11:48, 11 January 2015
Contents |
Introduction
Odorant-binding protein (OBP) are soluble proteins which involve in the processes of odorant detection in the olfactory sensilla.
The first OBP that was identified is Bovine odorant binding protein, that was isolated from a cow's mucus ref. Though functunaly same, vertebrates and insects OBP have different origin and stucture. OBPs are important for insect olfaction. For instance, OBP76a (LUSH) in the fly Drosophila melanogaster is required for the detection of the pheromone vaccenyl acetate [Ha and Smith, 2006; Xu et al., 2005] and has been proven to adopt a conformation that activates the odorant receptor [Laughlin et al., 2008].
OBP in insects
OBP Function
Despite five decades of intensive research, the exact roles of OBP and the mechanism by which the odorant receptor (OR) is activated are still in dispute [1][2].
A few functions have been suggested for OBP: 1. Solubelizing the odorant molecule and its transportation in the sensillar lymph.
2. Protecting the odorant molecule from the odorant degrading enzymes, in the sensillar lymph.
3. Activating of the odorant receptor on the dendrite membrane, by the odorant-OBP complex.
4. Mediating the deactivation of the odorant molecule after the activation of the receptor.
5. An organic anion (the protein has 9 negative charges).
Of all, the first role of OBP as an odorant solubilizer and carrier is generally accepted.
In order to explain the structure and function of these fascinating proteins, this page will further focus on a particular OBP - the well investigated Bombyx mori PBP: BmorPBP.
Bombyx mori BmorPBP (lets talk about sex..)
| |||||||||||
See also
References
Proteopedia Page Contributors and Editors (what is this?)
Nurit Eliash, Michal Harel, Joel L. Sussman, Alexander Berchansky, Jaime Prilusky