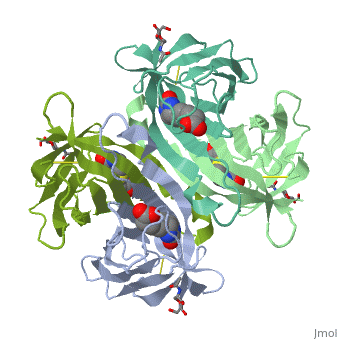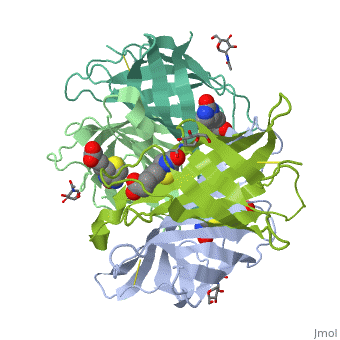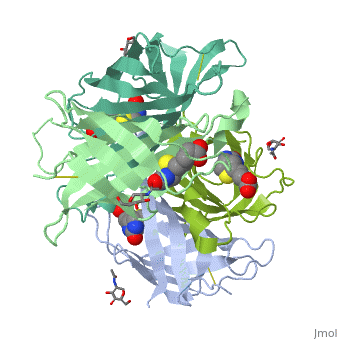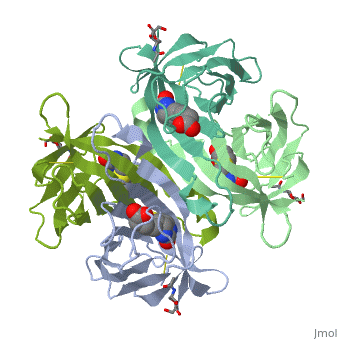User:Noam Gonen/Avidin
From Proteopedia
(Difference between revisions)
| Line 7: | Line 7: | ||
The tetrameric protein contains four identical subunits (homotetramer), each of which can bind to biotin (Vitamin B7, vitamin H) with a high degree of affinity and specificity. The dissociation constant of avidin is measured to be KD ≈ 10−15 M, making it one of the strongest known non-covalent bonds[2].The overall fold of the avidin monomer is constructed of eight antiparallel β-strands which form classical β-barrel. | The tetrameric protein contains four identical subunits (homotetramer), each of which can bind to biotin (Vitamin B7, vitamin H) with a high degree of affinity and specificity. The dissociation constant of avidin is measured to be KD ≈ 10−15 M, making it one of the strongest known non-covalent bonds[2].The overall fold of the avidin monomer is constructed of eight antiparallel β-strands which form classical β-barrel. | ||
== Monomer- monomer interaction == | == Monomer- monomer interaction == | ||
| - | '''Interaction 1-2''' | + | |
| + | '''Interaction 1-2''' | ||
| + | |||
Monomers are linked by hydrogen bond interactions between the respective N-terminal portions of β8-strands of each monomer. The β8-strands form a short antiparallel β-sheet. Each monomer contributes Trp-110 to its partner as an additional and very significant component of the biotin-binding site. When biotin is bound, interaction 1-2 is enhanced greatly, owing to the Trp-110-biotin interaction. The buried surface area of interaction is〖729Å〗^2. | Monomers are linked by hydrogen bond interactions between the respective N-terminal portions of β8-strands of each monomer. The β8-strands form a short antiparallel β-sheet. Each monomer contributes Trp-110 to its partner as an additional and very significant component of the biotin-binding site. When biotin is bound, interaction 1-2 is enhanced greatly, owing to the Trp-110-biotin interaction. The buried surface area of interaction is〖729Å〗^2. | ||
| + | |||
'''Interaction 1-3''' | '''Interaction 1-3''' | ||
| + | |||
Monomers interaction is relatively weak, involving only three equivalent hydrophobic residues from each monomer, Met-96, Val-115, and Ile-11. Resultant van der Waals interactions have the least contribution to the overall stability of the tetrameric structure of avidin. The buried surface area of interaction is〖120Å〗^2 . | Monomers interaction is relatively weak, involving only three equivalent hydrophobic residues from each monomer, Met-96, Val-115, and Ile-11. Resultant van der Waals interactions have the least contribution to the overall stability of the tetrameric structure of avidin. The buried surface area of interaction is〖120Å〗^2 . | ||
Revision as of 15:55, 17 January 2015
INTRODUCTION
Avidin is a tetrameric or dimeric[1] biotin-binding protein produced in the oviducts of birds, reptiles and amphibians and deposited in the whites of their eggs.
| |||||||||||