This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1058
From Proteopedia
(Difference between revisions)
| Line 6: | Line 6: | ||
==Isocitrate Lyase== | ==Isocitrate Lyase== | ||
===Structure=== | ===Structure=== | ||
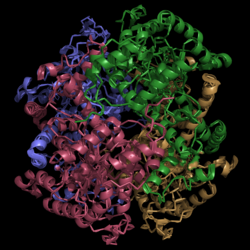
| - | [[Image:Normal_Crystal_Structure.png|250 px|center|thumb|'''Figure | + | [[Image:Normal_Crystal_Structure.png|250 px|center|thumb|'''Figure 1. Crystal Structure of Isocitrate Lyase.''' Quaternary structure is comprised of four subunits forming an alpha/beta barrel.]] |
| - | Isocitrate lyase | + | [http://www.rcsb.org/pdb/explore/explore.do?structureId=1f8i Isocitrate lyase] is a tetramer with 222 symmetry. Each subunit is composed of 14 alpha helices and 14 beta sheets which includes a total of 426 residues. These alpha helices and beta sheets form an unusual alpha/beta barrel seen in Figure 1. |
===Helix Swapping=== | ===Helix Swapping=== | ||
A unique structural feature of this enzyme is a phenomenon called "<scene name='69/694225/Helix_swapping/1'>helix swapping</scene>". | A unique structural feature of this enzyme is a phenomenon called "<scene name='69/694225/Helix_swapping/1'>helix swapping</scene>". | ||
| Line 19: | Line 19: | ||
==Mechanism of Action== | ==Mechanism of Action== | ||
| - | [[Image:TCA_Cycle.png|500 px|left|thumb|'''Figure | + | [[Image:TCA_Cycle.png|500 px|left|thumb|'''Figure 2. Citric Acid Cycle with Glyoxylate Shunt Pathway.''' In several bacterial species, there is a carbon conserving gloxylate shunt pathway that converts isocitrate to malate in two steps instead of the usual five steps.]] |
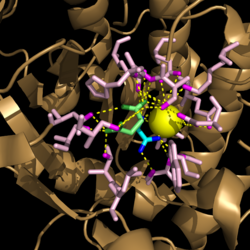
[[Image:Active_Site_Hydrogen_Bonding.png|250 px|left|thumb|'''Figure 3. Active site residues hydrogen bound to a cofactor and the products of the catalyzed isocitrate reaction.''' Glyoxylate is shown in blue, succinate is shown in green, and the Mg<sup>2+</sup> cofactor is shown in yellow.]] | [[Image:Active_Site_Hydrogen_Bonding.png|250 px|left|thumb|'''Figure 3. Active site residues hydrogen bound to a cofactor and the products of the catalyzed isocitrate reaction.''' Glyoxylate is shown in blue, succinate is shown in green, and the Mg<sup>2+</sup> cofactor is shown in yellow.]] | ||
==Disease Association== | ==Disease Association== | ||
Revision as of 19:06, 7 April 2015
Isocitrate Lyase from Mycobacterium tuberculosis
| |||||||||||