User:Braden Sciarra/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
== Active Site == | == Active Site == | ||
[[Image:Serine_with_active_site.png|400 px|right|thumb|Figure 3: Highly ordered hydrogen bonding network within the active site of ICL]] | [[Image:Serine_with_active_site.png|400 px|right|thumb|Figure 3: Highly ordered hydrogen bonding network within the active site of ICL]] | ||
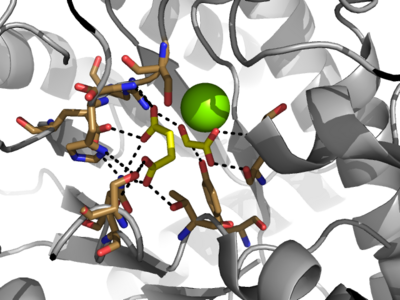
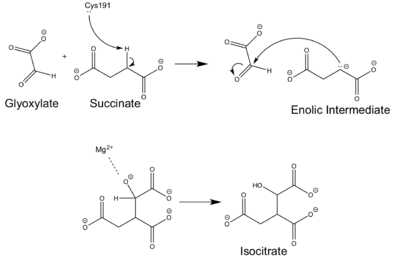
| - | The <scene name='69/697526/Binding_pocket/1'>Active Site</scene> of isocitrate lyase lies near the C-terminal ends of the Beta-strands of the active site. The glyoxylate substrate is held into place by a network of hydrogen bonds with Ser 91, Gly 92, Trp 93, and Arg 228<ref name="ICL">PMID:10932251</ref>>. The magnesium ion serves to stabilize the partial negative charge placed on the carbonyl oxygens of the glyoxylate. The other substrate, succinate, contains two carboxyl groups and possesses a similar network of hydrogen bonding that holds the ligand in place within the active site. One carboxylate group is hydrogen bound to Asn 313, Glu 295, Arg 228, and Gly 192. The second carboxylate is hydrogen bound to Thr 347, Asn 313, Ser 315, Ser 317, and His 193<ref name="ICL">PMID:10932251</ref>. The ordered hydrogen bonding within the active site orients the succinate molecule such that the alpha carbon is only 3.2 angstroms away from the deprotonated thiol group of Cys 191<ref name="ICL">PMID:10932251</ref><ref name="claisen" | + | The <scene name='69/697526/Binding_pocket/1'>Active Site</scene> of isocitrate lyase lies near the C-terminal ends of the Beta-strands of the active site. The glyoxylate substrate is held into place by a network of hydrogen bonds with Ser 91, Gly 92, Trp 93, and Arg 228<ref name="ICL">PMID:10932251</ref>>. The magnesium ion serves to stabilize the partial negative charge placed on the carbonyl oxygens of the glyoxylate. The other substrate, succinate, contains two carboxyl groups and possesses a similar network of hydrogen bonding that holds the ligand in place within the active site. One carboxylate group is hydrogen bound to Asn 313, Glu 295, Arg 228, and Gly 192. The second carboxylate is hydrogen bound to Thr 347, Asn 313, Ser 315, Ser 317, and His 193<ref name="ICL">PMID:10932251</ref>. The ordered hydrogen bonding within the active site orients the succinate molecule such that the alpha carbon is only 3.2 angstroms away from the deprotonated thiol group of Cys 191<ref name="ICL">PMID:10932251</ref><ref name="claisen">Masamune et al. Bio-Claisen condensation catalyzed by thiolase from Zoogloea ramigera. Active site cysteine residues. "Journal of the American Chemical Society" 111: 1879-1881 (1989). DOI: 10.1021/ja00187a053</ref>. |
===Catalytic Loop=== | ===Catalytic Loop=== | ||
Revision as of 00:27, 8 April 2015
Isocitrate Lyase from Mycobacterium Tuberculosis
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Sharma V, Sharma S, Hoener zu Bentrup K, McKinney JD, Russell DG, Jacobs WR Jr, Sacchettini JC. Structure of isocitrate lyase, a persistence factor of Mycobacterium tuberculosis. Nat Struct Biol. 2000 Aug;7(8):663-8. PMID:10932251 doi:10.1038/77964
- ↑ 2.0 2.1 2.2 2.3 Masamune et al. Bio-Claisen condensation catalyzed by thiolase from Zoogloea ramigera. Active site cysteine residues. "Journal of the American Chemical Society" 111: 1879-1881 (1989). DOI: 10.1021/ja00187a053