This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1058
From Proteopedia
(Difference between revisions)
| Line 23: | Line 23: | ||
===Catalytic Loop=== | ===Catalytic Loop=== | ||
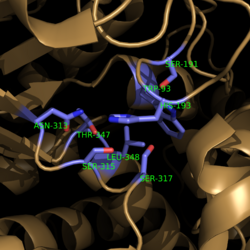
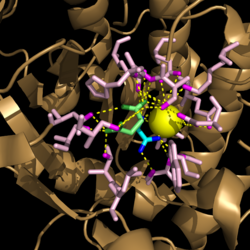
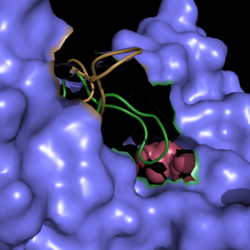
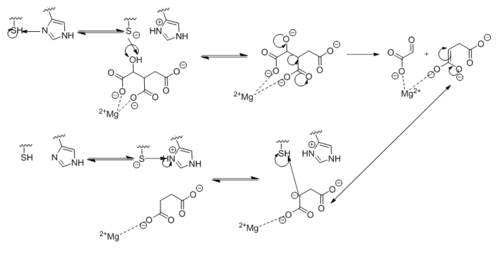
| - | [[Image:Active Loop Shift.png|250 px|left|thumb|'''Figure 4. Active Site Loop Shift.''' Binding of the ligand to the enzyme results in a conformational shift that facilitates the breakdown of isocitrate. The active site loop unbound is shown in wheat and the active site loop bound is shown in green.]] The catalytic loop of isocitrate lyase consists of residues 185-196 ('''Figure 4'''). The two most important are Cys191 and His193 as these form a charge relay strong enough to extract a proton from isocitrate. Poor electron density has been observed for residues His193 and Leu194 indicating that this loop is very flexible. <ref name="sharma"> Sharma, V.; Sharma, S.; Hoener zu Bentrup, K.; McKinney, J.; Russell, D.; ''et. al''; Structure of isocitrate lyase, a persistence factor of ''Mycobacterium tuberculosis''. ''Nat. Struct. Biol.''. '''2000'''. ''7(8)'':663-668. </ref> This data backs up the claim that that monomers of the protein are in a structural equilibria between the open and closed forms of the active site. In order for the catalytic loop to shift into the closed position necessary for catalysis, isocitrate must be within the binding pocket. The hydrogen bonding opportunities formed cause a ripple effect that shifts the catalytic loop into a closer position. <ref name="sharma"> Sharma, V.; Sharma, S.; Hoener zu Bentrup, K.; McKinney, J.; Russell, D.; ''et. al''; Structure of isocitrate lyase, a persistence factor of ''Mycobacterium tuberculosis''. ''Nat. Struct. Biol.''. '''2000'''. ''7(8)'':663-668. </ref> This shift also causes the C-terminal domain of the subunit (residues 411-428) to <scene name='69/694225/C-terminus_loop_in_cat_loop/1'>move into the former position of the catalytic loop (in green)</scene>. The C-terminal domain is stabilized by an <scene name='69/694225/Lys_electrostatic/1'>electrostatic interaction</scene> with Lys189. This combined movement locks the active site residues into a proper orientation for lysis of a C-C bond within isocitrate. <ref name="sharma"> Sharma, V.; Sharma, S.; Hoener zu Bentrup, K.; McKinney, J.; Russell, D.; ''et. al''; Structure of isocitrate lyase, a persistence factor of ''Mycobacterium tuberculosis''. ''Nat. Struct. Biol.''. '''2000'''. ''7(8)'':663-668. </ref> | + | [[Image:Active Loop Shift.png|250 px|left|thumb|'''Figure 4. Active Site Loop Shift.''' Binding of the ligand to the enzyme results in a conformational shift that facilitates the breakdown of isocitrate. The active site loop unbound is shown in wheat and the active site loop bound is shown in green.]] The catalytic loop of isocitrate lyase consists of residues 185-196 ('''Figure 4'''). The two most important are Cys191 and His193 as these form a charge relay strong enough to extract a proton from isocitrate. Poor electron density has been observed for residues His193 and Leu194 indicating that this loop is very flexible. <ref name="sharma"> Sharma, V.; Sharma, S.; Hoener zu Bentrup, K.; McKinney, J.; Russell, D.; ''et. al''; Structure of isocitrate lyase, a persistence factor of ''Mycobacterium tuberculosis''. ''Nat. Struct. Biol.''. '''2000'''. ''7(8)'':663-668. </ref> This data backs up the claim that that monomers of the protein are in a structural equilibria between the open and closed forms of the active site. In order for the catalytic loop to shift into the closed position necessary for catalysis, isocitrate must be within the binding pocket. The hydrogen bonding opportunities formed cause a ripple effect that shifts the catalytic loop into a closer position. <ref name="sharma"> Sharma, V.; Sharma, S.; Hoener zu Bentrup, K.; McKinney, J.; Russell, D.; ''et. al''; Structure of isocitrate lyase, a persistence factor of ''Mycobacterium tuberculosis''. ''Nat. Struct. Biol.''. '''2000'''. ''7(8)'':663-668. </ref> This shift also causes the C-terminal domain of the subunit (residues 411-428) to <scene name='69/694225/C-terminus_loop_in_cat_loop/1'>move into the former position of the catalytic loop (in green)</scene>. The C-terminal domain is then stabilized by an <scene name='69/694225/Lys_electrostatic/1'>electrostatic interaction</scene> with Lys189. This combined movement locks the active site residues into a proper orientation for lysis of a C-C bond within isocitrate. <ref name="sharma"> Sharma, V.; Sharma, S.; Hoener zu Bentrup, K.; McKinney, J.; Russell, D.; ''et. al''; Structure of isocitrate lyase, a persistence factor of ''Mycobacterium tuberculosis''. ''Nat. Struct. Biol.''. '''2000'''. ''7(8)'':663-668. </ref> |
Revision as of 16:40, 10 April 2015
Isocitrate Lyase from Mycobacterium tuberculosis
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Sharma, V.; Sharma, S.; Hoener zu Bentrup, K.; McKinney, J.; Russell, D.; et. al; Structure of isocitrate lyase, a persistence factor of Mycobacterium tuberculosis. Nat. Struct. Biol.. 2000. 7(8):663-668.
- ↑ Gould, T.; van de Langemheen, H.; Muñoz-Elías, E.; McKinney, D.; Sacchettini, J.; Dual role of isocitrate lyase 1 in the glyoxylate and methylcitrate cycles in Mycobacterium tuberculosis. Molecular Microbiology. 2006. 61(4):940-947. doi:10.1111/j.1365-2958.2006.05297.x.
- ↑ Cozzone, A.; Regulation of acetate metabolism by protein phosphorylation in enteric bacteria. Annual Review of Microbiology. 1998, 52:127-164. doi: 10.1146/annurev.micro.52.1.127.
- ↑ Srivastava, V.; Janin, A.; Srivastava, B.; Srivastava, R.; Selection of genes of Mycobacterium tuberculosis upregulated during residence in lungs of infected mice. ScienceDirect. 2007. doi:10.1016/j.tube.2007.10.002.
- ↑ Muñoz-Elías, E.; McKinney, J.; M. tuberculosis isocitrate lyases 1 and 2 are jointly required for in vivo growth and virulence. Nat. Med. 2005. 11(6):638-644. doi:10.1038/nm1252.
- ↑ Dunn, M.; Ramírez-Trujillo, J.; Hernández-Lucas, I.; Major roles of isocitrate lyase and malate synthase in bacterial and fungal pathogenesis. Microbiology. 2009. 155:3166-3175. doi:10.1099/mic.0.030858-0.