This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1070
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
<StructureSection load='2lqj' size='340' side='right' caption='C-terminal Domain of Mg2+ transport P-type ATPase C (PDB: [http://www.rcsb.org/pdb/explore.do?structureId=2lqj 2LQJ])' scene='69/698113/Rainbow-colored_spectrum/2'> | <StructureSection load='2lqj' size='340' side='right' caption='C-terminal Domain of Mg2+ transport P-type ATPase C (PDB: [http://www.rcsb.org/pdb/explore.do?structureId=2lqj 2LQJ])' scene='69/698113/Rainbow-colored_spectrum/2'> | ||
==Introduction== | ==Introduction== | ||
| - | [http://en.wikipedia.org/wiki/Tuberculosis Tuberculosis], caused by ''[http://en.wikipedia.org/wiki/Mycobacterium_tuberculosis Mycobacterium tuberculosis]'', is a [http://en.wikipedia.org/wiki/Respiratory_tract_infection respiratory infection] still prevalent throughout the world. During the last decade, the emergence of [http://en.wikipedia.org/wiki/Multiple_drug_resistance multi-drug resistant] strains of ''M. tuberculosis'' has given rise to the need for the development of new antibiotics in order to combat the infection<ref>Singh, G.; Singh, G.; Jadeja, D.; Kaur, J. Lipid hydrolyzing enzymes in virulence: Mycobacterium tuberculosis as a model system. Critical Reviews in Microbiology 2010, 36(3): 259-269. DOI: 10.3109/1040841X.2010.482923.</ref>. In order to develop an efficacious antibiotic, the drug must be able to target a unique aspect of the bacteria, such as a protein, that is critical for its full virulence and survival. MgtC, an [http://en.wikipedia.org/wiki/Integral_membrane_protein integral protein] embedded in the extracellular membrane of ''M. tuberculosis'', has recently been hypothesized as a novel drug target to resolve tuberculosis infections. The targeting of MgtC was a result of observing that upon deletion of the protein from ''M. tuberculosis'', the bacteria are no longer able to survive due to inhibition of [http://en.wiktionary.org/wiki/intramacrophage intramacrophage] growth. <ref name="mgtc">Yang, Y.; Labesse, G.; Carrere-Kremer, S.; Esteves, K.; Kremer, L.; Cohen-Gonsaud, M.; Blanc-Potard, A. The C-terminal domain of the virulence factor mgtc is a divergent act domain. J Bacteriol. 2012, 194(22): 6255-6263. DOI: 10.1128/JB.01424-12.</ref>. | + | [http://en.wikipedia.org/wiki/Tuberculosis Tuberculosis], caused by ''[http://en.wikipedia.org/wiki/Mycobacterium_tuberculosis Mycobacterium tuberculosis]'', is a [http://en.wikipedia.org/wiki/Respiratory_tract_infection respiratory infection] still prevalent throughout the world. During the last decade, the emergence of [http://en.wikipedia.org/wiki/Multiple_drug_resistance multi-drug resistant] strains of ''M. tuberculosis'' has given rise to the need for the development of new [http://en.wikipedia.org/wiki/Antibiotics antibiotics] in order to combat the infection<ref>Singh, G.; Singh, G.; Jadeja, D.; Kaur, J. Lipid hydrolyzing enzymes in virulence: Mycobacterium tuberculosis as a model system. Critical Reviews in Microbiology 2010, 36(3): 259-269. DOI: 10.3109/1040841X.2010.482923.</ref>. In order to develop an efficacious antibiotic, the drug must be able to target a unique aspect of the bacteria, such as a protein, that is critical for its full virulence and survival. MgtC, an [http://en.wikipedia.org/wiki/Integral_membrane_protein integral protein] embedded in the extracellular membrane of ''M. tuberculosis'', has recently been hypothesized as a novel drug target to resolve tuberculosis infections. The targeting of MgtC was a result of observing that upon deletion of the protein from ''M. tuberculosis'', the bacteria are no longer able to survive due to inhibition of [http://en.wiktionary.org/wiki/intramacrophage intramacrophage] growth. <ref name="mgtc">Yang, Y.; Labesse, G.; Carrere-Kremer, S.; Esteves, K.; Kremer, L.; Cohen-Gonsaud, M.; Blanc-Potard, A. The C-terminal domain of the virulence factor mgtc is a divergent act domain. J Bacteriol. 2012, 194(22): 6255-6263. DOI: 10.1128/JB.01424-12.</ref>. |
== Structure == | == Structure == | ||
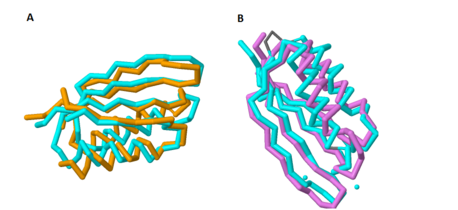
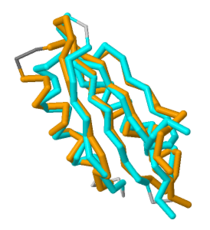
Based on its [http://en.wikipedia.org/wiki/Protein_tertiary_structure tertiary structure], this protein has been placed into a larger group of proteins known as the [http://www.ncbi.nlm.nih.gov/Structure/cdd/cddsrv.cgi?uid=120498 MgtC superfamily]. The overall structure of MgtC is constituted by two [http://en.wikipedia.org/wiki/Protein_domain domains]: an N-terminal domain and a C-terminal domain. Each of these domains have striking similarities and differences with other MgtC-like proteins.<ref name="mgtc"/> | Based on its [http://en.wikipedia.org/wiki/Protein_tertiary_structure tertiary structure], this protein has been placed into a larger group of proteins known as the [http://www.ncbi.nlm.nih.gov/Structure/cdd/cddsrv.cgi?uid=120498 MgtC superfamily]. The overall structure of MgtC is constituted by two [http://en.wikipedia.org/wiki/Protein_domain domains]: an N-terminal domain and a C-terminal domain. Each of these domains have striking similarities and differences with other MgtC-like proteins.<ref name="mgtc"/> | ||
| Line 83: | Line 83: | ||
==Clinical Relevance == | ==Clinical Relevance == | ||
| - | The development of an antibiotic which targets and inhibits MgtC could come from exploitation and enhancement of the process which promotes its degradation within ''Mycobacterium tuberculosis.'' MgtR, a hydrophobic peptide, promotes the degradation of MgtC upon high expression within the bacteria.<ref name="mgtr"/> As previously stated, inadequate levels of MgtC within ''M. tuberculosis'' results in an inability to | + | The development of an antibiotic which targets and inhibits MgtC could come from exploitation and enhancement of the process which promotes its degradation within ''Mycobacterium tuberculosis.'' MgtR, a hydrophobic peptide, promotes the degradation of MgtC upon high expression within the bacteria.<ref name="mgtr"/> As previously stated, inadequate levels of MgtC within ''M. tuberculosis'' results in an inability to grow and survive. <ref name="mgtr"/> It is quite reasonable that analogues of MgtR could be developed, injected ([http://en.wikipedia.org/wiki/Subcutaneous_injection subcutaneously]) into infected patients, and resolve the tuberculosis infection by promoting degradation of MgtC and impairing growth of ''M. tuberculosis.'' |
==Future Work== | ==Future Work== | ||
Revision as of 13:47, 14 April 2015
| This Sandbox is Reserved from 02/09/2015, through 05/31/2016 for use in the course "CH462: Biochemistry 2" taught by Geoffrey C. Hoops at the Butler University. This reservation includes Sandbox Reserved 1051 through Sandbox Reserved 1080. |
To get started:
More help: Help:Editing |
MgtC: A Virulence Factor From Mycobacterium tuberculosis
| |||||||||||
References
- ↑ Singh, G.; Singh, G.; Jadeja, D.; Kaur, J. Lipid hydrolyzing enzymes in virulence: Mycobacterium tuberculosis as a model system. Critical Reviews in Microbiology 2010, 36(3): 259-269. DOI: 10.3109/1040841X.2010.482923.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 Yang, Y.; Labesse, G.; Carrere-Kremer, S.; Esteves, K.; Kremer, L.; Cohen-Gonsaud, M.; Blanc-Potard, A. The C-terminal domain of the virulence factor mgtc is a divergent act domain. J Bacteriol. 2012, 194(22): 6255-6263. DOI: 10.1128/JB.01424-12.
- ↑ Blanc-Potard, A.B.; Lafay, B. MgtC as a horizontally-acquired virulence factor of intracellular bacterial pathogens : evidence from molecular phylogeny and comparative genomics. J Mol Evol. 2003, 57(4): 479-86. DOI: 10.1007/s00239-003-2496-4
- ↑ Belon, C.; Gannoun-Zaki, L.; Lutfalla, G.; Kremer, L.; Blanc-Potard, A.B. Mycobacterium marinum mgtc plays a role in phagocytosis but is dispensable for intracellular multiplication. Plos One 2014, 1-23. DOI: 10.1371/journal.pone.0116052.
- ↑ 5.0 5.1 5.2 Jean-Francois, F.L.; Dai, J.; Yu, L. ; Myrick, A. ; Rubin, E. ; et al. Binding of mgtr, a salmonella transmembrane regulatory peptide, to mgtc, a mycobacterium tuberculosis virulence factor: a structural study. DOI:10.1016/j.jmb.2013.10.014