This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1087
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
| - | <StructureSection load='1iwg' size='450' side='right' scene='Insert optional scene name here' caption=' | + | <StructureSection load='1iwg' size='450' side='right' scene='Insert optional scene name here' caption='AcrB transporter ([[1iwg]])' >__NOTOC__ |
{{UniHelsinki_ProteinCourse_2015}} | {{UniHelsinki_ProteinCourse_2015}} | ||
| Line 51: | Line 51: | ||
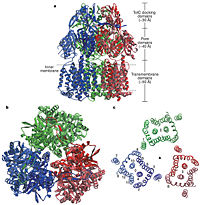
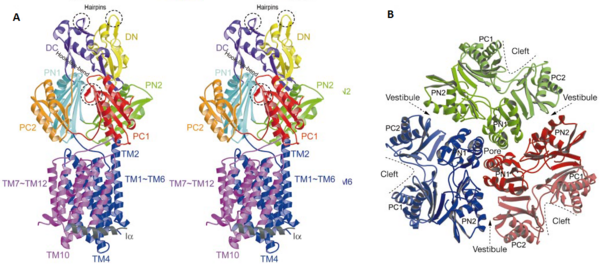
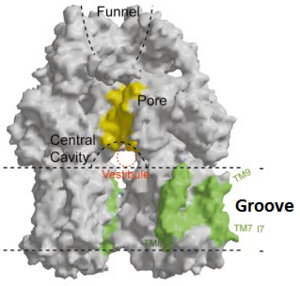
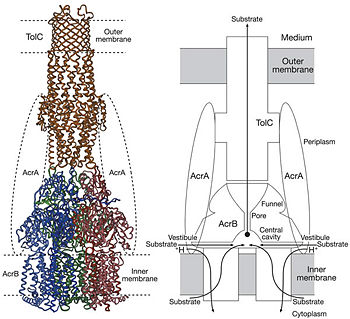
Twelve α-helices of each monomer forms the transmembrane domain. Six α-helices in the N-terminal and the six in C-terminal are arranged symmetrically. These helices are long and they reach outside the cytoplasmic surface of the membrane. The membrane domain contains an additional extra-membrane a-helix (Ia) located between <scene name='70/700000/Acrb_tm_6/2'>TM6</scene> and <scene name='70/700000/Acrb_tm7/2'>TM7</scene> attached to the cytoplasmic membrane surface. Between <scene name='70/700000/Acrb_tm8/3'>TM8</scene> and TM7 locates a groove within the transmembrane domain of each monomer. Amino acid residues 860-868 in TM8 are in a disordered state. Through the disordered region of the top of TM8, the groove is connected with the cavity. This domain contains three functionally critical residues: Asp407, Asp408 and Lys940. The residues are shown in this <scene name='70/700000/Acrb_residues/2'>link</scene>, Asp407 as pink, Asp408 as green and Lys940 as blue. When these are mutated the whole complex loses its drug resistance.<ref name= "Nakashima"/> | Twelve α-helices of each monomer forms the transmembrane domain. Six α-helices in the N-terminal and the six in C-terminal are arranged symmetrically. These helices are long and they reach outside the cytoplasmic surface of the membrane. The membrane domain contains an additional extra-membrane a-helix (Ia) located between <scene name='70/700000/Acrb_tm_6/2'>TM6</scene> and <scene name='70/700000/Acrb_tm7/2'>TM7</scene> attached to the cytoplasmic membrane surface. Between <scene name='70/700000/Acrb_tm8/3'>TM8</scene> and TM7 locates a groove within the transmembrane domain of each monomer. Amino acid residues 860-868 in TM8 are in a disordered state. Through the disordered region of the top of TM8, the groove is connected with the cavity. This domain contains three functionally critical residues: Asp407, Asp408 and Lys940. The residues are shown in this <scene name='70/700000/Acrb_residues/2'>link</scene>, Asp407 as pink, Asp408 as green and Lys940 as blue. When these are mutated the whole complex loses its drug resistance.<ref name= "Nakashima"/> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| Line 74: | Line 60: | ||
Studies have been done on the substrate binding and specificity and the periplasmic part of the tripartite efflux system is found important to the substrate specificity. In the study on antibiotics to AcrB, Phe<scene name='70/700000/Acrb_386/1'>386</scene> (TM3) was reported as one the main hydrophobic contacts. However, currently, the theoretical explanation of the wide variety of substrates is still lacking.<ref name= "Nakashima"/> | Studies have been done on the substrate binding and specificity and the periplasmic part of the tripartite efflux system is found important to the substrate specificity. In the study on antibiotics to AcrB, Phe<scene name='70/700000/Acrb_386/1'>386</scene> (TM3) was reported as one the main hydrophobic contacts. However, currently, the theoretical explanation of the wide variety of substrates is still lacking.<ref name= "Nakashima"/> | ||
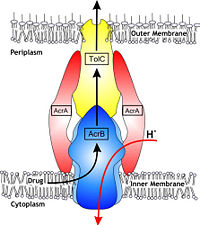
The tripartite efflux system coupled with proton-motive force across the cytoplasmic membrane. It is the binding and release of protons in the transmembrane domain that is crucial to the energy transduction.<ref name= "Klaas"/> Certain key residues had been identified as crucial to the protons translocation. They are the residues Lys940 (TM10) and Asp407 and 408 (TM4) harbored in <scene name='70/700000/Acrb_tm_4/2'>TM4</scene> and <scene name='70/700000/Acrb_tm10/2'>TM10</scene> in each monomer. When the ion pairs between them are disrupted due to the transient protonation of residues mentioned above, there may be conformational change of TM 4 and 10. Through possible remote conformational coupling, the conformational change of TM 4 and 10 may induce the opening of the pore.<ref name= "Nakashima"/> | The tripartite efflux system coupled with proton-motive force across the cytoplasmic membrane. It is the binding and release of protons in the transmembrane domain that is crucial to the energy transduction.<ref name= "Klaas"/> Certain key residues had been identified as crucial to the protons translocation. They are the residues Lys940 (TM10) and Asp407 and 408 (TM4) harbored in <scene name='70/700000/Acrb_tm_4/2'>TM4</scene> and <scene name='70/700000/Acrb_tm10/2'>TM10</scene> in each monomer. When the ion pairs between them are disrupted due to the transient protonation of residues mentioned above, there may be conformational change of TM 4 and 10. Through possible remote conformational coupling, the conformational change of TM 4 and 10 may induce the opening of the pore.<ref name= "Nakashima"/> | ||
| - | |||
| - | |||
| - | |||
Revision as of 13:20, 23 April 2015
| |||||||||||