This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1058
From Proteopedia
(Difference between revisions)
| Line 27: | Line 27: | ||
===Regulation=== | ===Regulation=== | ||
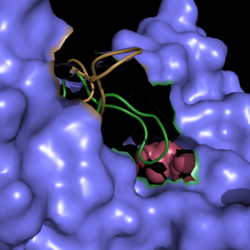
| - | Isocitrate lyase competes with [http://en.wikipedia.org/wiki/Isocitrate_dehydrogenase isocitrate dehydrogenase], an enzyme found in the [http://en.wikipedia.org/wiki/Citric_acid_cycle citric cycle], for isocitrate processing. The favoritism of one enzyme over the other is controlled by the phosphorylation of isocitrate dehydrogenase. This enzyme has a much higher affinity for isocitrate as compared to isocitrate lyase. Phosphorylation of isocitrate dehydrogenase inactivates the enzyme and leades to increased isocitrate lyase activity. <ref name="cozzone"> Cozzone, A.; Regulation of acetate metabolism by protein phosphorylation in enteric bacteria. ''Annual Review of Microbiology''. '''1998''', ''52'':127-164. doi: 10.1146/annurev.micro.52.1.127. </ref> | + | <scene name='69/694225/Isocitrate_lyase/1'>Isocitrate lyase</scene> competes with [http://en.wikipedia.org/wiki/Isocitrate_dehydrogenase isocitrate dehydrogenase], an enzyme found in the [http://en.wikipedia.org/wiki/Citric_acid_cycle citric cycle], for isocitrate processing. The favoritism of one enzyme over the other is controlled by the phosphorylation of isocitrate dehydrogenase. This enzyme has a much higher affinity for isocitrate as compared to isocitrate lyase. Phosphorylation of isocitrate dehydrogenase inactivates the enzyme and leades to increased isocitrate lyase activity. <ref name="cozzone"> Cozzone, A.; Regulation of acetate metabolism by protein phosphorylation in enteric bacteria. ''Annual Review of Microbiology''. '''1998''', ''52'':127-164. doi: 10.1146/annurev.micro.52.1.127. </ref> |
| Line 34: | Line 34: | ||
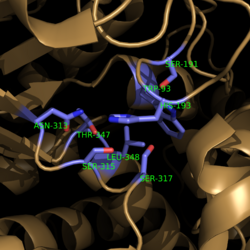
==Mechanism of Action== | ==Mechanism of Action== | ||
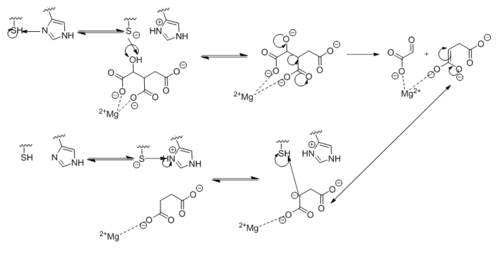
[[Image:Complete_Mechanism.PNG|500 px|right|thumb|'''Figure 5. Observed Mechanism for the Breakdown of Isocitrate by Isocitrate Lyase.''']] | [[Image:Complete_Mechanism.PNG|500 px|right|thumb|'''Figure 5. Observed Mechanism for the Breakdown of Isocitrate by Isocitrate Lyase.''']] | ||
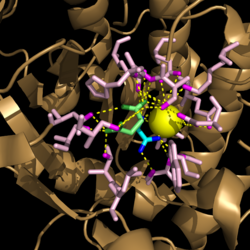
| - | Within | + | Within <scene name='69/694225/Isocitrate_lyase/3'>Isocitrate lyase</scene>, His193 shifts the pKa of Cys191 and removes its proton. This allows Cys191 to extract a proton from the hydroxyl group of isocitrate. The resulting oxyanion forms a carbonyl and forces the lysis of a C-C bond. Glyoxylate and the enol form of succinate are formed and stabilized with a Mg<sup>2+</sup> ion. The succinate enolate resonates and extracts the proton back from Cys191 to form succinate ('''Figure 5'''). |
Revision as of 03:44, 24 April 2015
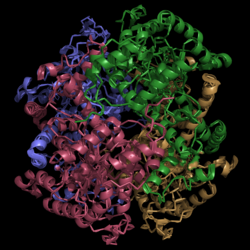
Isocitrate Lyase from Mycobacterium tuberculosis
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Sharma, V.; Sharma, S.; Hoener zu Bentrup, K.; McKinney, J.; Russell, D.; et. al; Structure of isocitrate lyase, a persistence factor of Mycobacterium tuberculosis. Nat. Struct. Biol.. 2000. 7(8):663-668.
- ↑ Gould, T.; van de Langemheen, H.; Muñoz-Elías, E.; McKinney, D.; Sacchettini, J.; Dual role of isocitrate lyase 1 in the glyoxylate and methylcitrate cycles in Mycobacterium tuberculosis. Molecular Microbiology. 2006. 61(4):940-947. doi:10.1111/j.1365-2958.2006.05297.x.
- ↑ Cozzone, A.; Regulation of acetate metabolism by protein phosphorylation in enteric bacteria. Annual Review of Microbiology. 1998, 52:127-164. doi: 10.1146/annurev.micro.52.1.127.
- ↑ Muñoz-Elías, E.; McKinney, J.; M. tuberculosis isocitrate lyases 1 and 2 are jointly required for in vivo growth and virulence. Nat. Med. 2005. 11(6):638-644. doi:10.1038/nm1252.
- ↑ Srivastava, V.; Janin, A.; Srivastava, B.; Srivastava, R.; Selection of genes of Mycobacterium tuberculosis upregulated during residence in lungs of infected mice. ScienceDirect. 2007. doi:10.1016/j.tube.2007.10.002.
- ↑ Dunn, M.; Ramírez-Trujillo, J.; Hernández-Lucas, I.; Major roles of isocitrate lyase and malate synthase in bacterial and fungal pathogenesis. Microbiology. 2009. 155:3166-3175. doi:10.1099/mic.0.030858-0.