This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1068
From Proteopedia
| Line 5: | Line 5: | ||
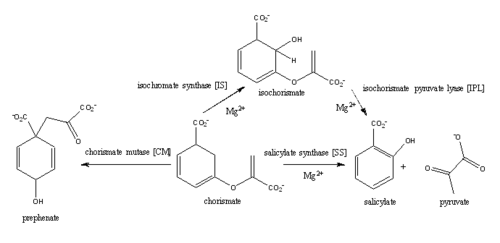
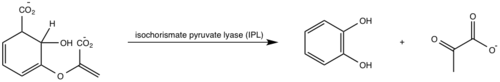
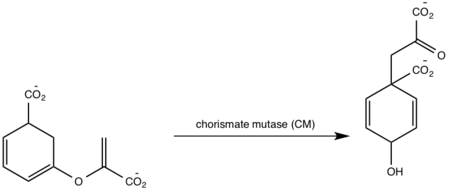
<scene name='69/694235/3log/12'>Salicylate synthase</scene> from [http://en.wikipedia.org/wiki/''Mycobacterium_tuberculosis''] (MtbI) is a highly promiscuous enzyme that has four distinct activities ''in vivo'': [http://en.wikipedia.org/wiki/Isochorismate_synthase isochorismate synthase] (IS), [http://www.proteopedia.org/wiki/index.php/Isochorismate_pyruvate_lyase isochorismate pyruvate lyase] (IPL), [http://www.rcsb.org/pdb/results/results.do?outformat=&qrid=8A8773E9&tabtoshow=Current salicylate synthase] (SS) and [http://en.wikipedia.org/wiki/Chorismate_mutase chorismate mutate] (CM)(Ferrer 2012). MtbI belongs to the chorismate-utilizing enzyme family, which consists of structural homologues (<scene name='69/694235/Irp9/5'>Ipr9</scene>, <scene name='69/694235/Menf/3'>MenF</scene>, <scene name='69/694235/Entc/3'>EntC</scene>, and <scene name='69/694235/Mbti/3'>MbtI</scene>) that isomerize chromate to isochorismate and share a fold of two α/β subdomains, each comprising of a antiparallel β-sheet with helices packed against it(Ferrer 2012, Lamb 2011). These enzymes are present in bacteria, fungi, plants and apicomplexan parasites and catalyze the initial reactions of menaquinone, siderophore, and tryptophan biosynthesis(ferrer 2012, Lamb 2011, Voss 1999). The IS, IPL, and SS activity of MbtI require the presence of a magnesium ion within the active site, while CM activity is only observed in absence of the magnesium cation(ferrer 2012). IS, IPL, and SS activity are also modulated by the pH of the medium(ferrer 2012). Isochorismate is the primary product at pH values below 7.5 and salicylate is the primary product formed at pH 8(ferrer 2012, Zwahlen 2006). | <scene name='69/694235/3log/12'>Salicylate synthase</scene> from [http://en.wikipedia.org/wiki/''Mycobacterium_tuberculosis''] (MtbI) is a highly promiscuous enzyme that has four distinct activities ''in vivo'': [http://en.wikipedia.org/wiki/Isochorismate_synthase isochorismate synthase] (IS), [http://www.proteopedia.org/wiki/index.php/Isochorismate_pyruvate_lyase isochorismate pyruvate lyase] (IPL), [http://www.rcsb.org/pdb/results/results.do?outformat=&qrid=8A8773E9&tabtoshow=Current salicylate synthase] (SS) and [http://en.wikipedia.org/wiki/Chorismate_mutase chorismate mutate] (CM)(Ferrer 2012). MtbI belongs to the chorismate-utilizing enzyme family, which consists of structural homologues (<scene name='69/694235/Irp9/5'>Ipr9</scene>, <scene name='69/694235/Menf/3'>MenF</scene>, <scene name='69/694235/Entc/3'>EntC</scene>, and <scene name='69/694235/Mbti/3'>MbtI</scene>) that isomerize chromate to isochorismate and share a fold of two α/β subdomains, each comprising of a antiparallel β-sheet with helices packed against it(Ferrer 2012, Lamb 2011). These enzymes are present in bacteria, fungi, plants and apicomplexan parasites and catalyze the initial reactions of menaquinone, siderophore, and tryptophan biosynthesis(ferrer 2012, Lamb 2011, Voss 1999). The IS, IPL, and SS activity of MbtI require the presence of a magnesium ion within the active site, while CM activity is only observed in absence of the magnesium cation(ferrer 2012). IS, IPL, and SS activity are also modulated by the pH of the medium(ferrer 2012). Isochorismate is the primary product at pH values below 7.5 and salicylate is the primary product formed at pH 8(ferrer 2012, Zwahlen 2006). | ||
| - | The salicylate synthase activity of MbtI catalyzes the first committed step in the synthesis of the iron chelating [http://en.wikipedia.org/wiki/Siderophore siderophore], mycobactin T, in ''Mycobacterium tuberculosis'' (Figure 1)<ref name= "5a">PMID:22607697</ref>. Mycobactin T is synthesized by the proteins encoded by the ''mbt'' and ''mbt2'' gene clusters <ref name="5a"/>. The gene Rv2386c is essential for the in vitro growth of ''M. tuberculosis'' and codes the enzyme MbtI (turvey, 2010). This complex secondary metabolite is essential for both virulence and survival of ''M. tuberculosis''(ferrer 2012, Voss 1999, Harrison 2006). Therefore, inhibitors of salicylate synthase may serve as potential TB therapies with a novel mode of action <ref name= "1a"> | + | The salicylate synthase activity of MbtI catalyzes the first committed step in the synthesis of the iron chelating [http://en.wikipedia.org/wiki/Siderophore siderophore], mycobactin T, in ''Mycobacterium tuberculosis'' (Figure 1)<ref name= "5a">PMID:22607697</ref>. Mycobactin T is synthesized by the proteins encoded by the ''mbt'' and ''mbt2'' gene clusters <ref name="5a"/>. The gene Rv2386c is essential for the in vitro growth of ''M. tuberculosis'' and codes the enzyme MbtI (turvey, 2010). This complex secondary metabolite is essential for both virulence and survival of ''M. tuberculosis''(ferrer 2012, Voss 1999, Harrison 2006). Therefore, inhibitors of salicylate synthase may serve as potential TB therapies with a novel mode of action <ref name= "1a"> PMID:20512795</ref> <ref name= "2a">PMID:23108268</ref> <ref name= "7a">Voss, James J., Kerry Rutter, Benjamin G. Schroedor, Hua Su, and YaQi Zhu. "The salicylate-derived mycobactin siderophores of Mycobacterium tuberculosis are essential for growth in macrophages." Proceedings of the National Academy of Sciences 97.3 (2000): 1252-57. Web. 14 Mar. 2015.</ref> <ref name= "5a"/> <ref name= "4a">DOI:10.1021/bi2009739</ref> <ref name= "9a">PMID:14982443</ref> |
[[Image:Pathways.png|500 px|center|thumb|'''Figure 1:''' Pathways catalyzed by wild-type MbtI<ref>PMID:22307014</ref>.]] | [[Image:Pathways.png|500 px|center|thumb|'''Figure 1:''' Pathways catalyzed by wild-type MbtI<ref>PMID:22307014</ref>.]] | ||
| Line 40: | Line 40: | ||
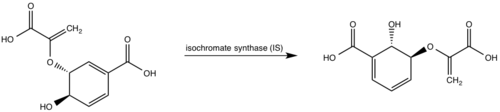
'''Isochorismate synthase (IS)''' | '''Isochorismate synthase (IS)''' | ||
| - | Currently, isochorismate is believed to be formed from chorismate through a proposed Sn2 mechanism involving nucleophilic attack of an activated water molecule to the C2 center followed by either a concerted or stepwise elimination of the C4 hydroxyl group <ref | + | Currently, isochorismate is believed to be formed from chorismate through a proposed Sn2 mechanism involving nucleophilic attack of an activated water molecule to the C2 center followed by either a concerted or stepwise elimination of the C4 hydroxyl group <ref name="9a"/>. Lys205 has been proposed to act as the catalytic base, activating a water molecule in the active site by abstracting one of its protons. However, mutational analysis of Lys205 suggested that the lysine reside is not the sole determinant in the activation of a water molecule for nucleophilic attack of the C2 center. Studies have shown that Lys205 is protonated at neutral pH and therefore can't act as a base to activate the water molecule, agreeing with the mutational analysis data. Instead of Lys205, Glu297 residue has been proposed to act as a base in the activation of the water molecule. The magnesium ion forces the negatively charged Glu297 residue to face toward the active site and the pKa of Glu297 (3.9) suggest an unprotonated state. Furthermore, Glu297 forms a hydrogen bond with a water molecule within the active site as well as with Lys205, which is in turn hydrogen bonded to C1 carboxylate group of chorismate and the oxygen of the nucleophilic water molecule. The glutamic residue (Gly252) could protonate the C4 leaving hydroxyl group. The pKa of Gly252 (7.7) suggest that is it is the only protonated glutamate residue in the active site at pH 7 and thus able to protonate the C4 leaving group. The pKa of Gly252 also accounts for the accumulation of isochorismate at pH values below 7.5. |
| Line 59: | Line 59: | ||
Mycobactin is a siderophore synthesized by the proteins encoded by the ''mbt'' and ''mbt2'' gene clusters <ref name="5a"/>. The gene Rv2386c is essential for the in vitro growth of "M. tuberculosis" and codes the enzyme MbtI. (turvey, 2010) | Mycobactin is a siderophore synthesized by the proteins encoded by the ''mbt'' and ''mbt2'' gene clusters <ref name="5a"/>. The gene Rv2386c is essential for the in vitro growth of "M. tuberculosis" and codes the enzyme MbtI. (turvey, 2010) | ||
| - | MbtI catalyses the first committed step in the biosynthesis of the siderophore mycobactin and is a potential target for inhibition. The salicylate synthase activity of MbtI produces salicylate and pyruvate from chorismate through an isochorismate intermediate. Inhibition of MbtI activity would decrease the production of salicylate and therefore the synthesis of mycobactin; leading to a decrease in iron acquisition and pathogenesis of ''M. tuberculosis''<ref | + | MbtI catalyses the first committed step in the biosynthesis of the siderophore mycobactin and is a potential target for inhibition. The salicylate synthase activity of MbtI produces salicylate and pyruvate from chorismate through an isochorismate intermediate. Inhibition of MbtI activity would decrease the production of salicylate and therefore the synthesis of mycobactin; leading to a decrease in iron acquisition and pathogenesis of ''M. tuberculosis''<ref name= "7a"/> . |
Revision as of 21:36, 26 April 2015
Contents |
Mycobacterium tuberculosis salicylate synthase (Mbt1)
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Chi G, Manos-Turvey A, O'Connor PD, Johnston JM, Evans GL, Baker EN, Payne RJ, Lott JS, Bulloch EM. Implications of Binding Mode and Active Site Flexibility for Inhibitor Potency against the Salicylate Synthase from Mycobacterium tuberculosis. Biochemistry. 2012 Jun 7. PMID:22607697 doi:10.1021/bi3002067
- ↑ 2.0 2.1 Manos-Turvey A, Bulloch EM, Rutledge PJ, Baker EN, Lott JS, Payne RJ. Inhibition studies of Mycobacterium tuberculosis salicylate synthase (MbtI). ChemMedChem. 2010 Jul 5;5(7):1067-79. PMID:20512795 doi:10.1002/cmdc.201000137
- ↑ 3.0 3.1 Manos-Turvey A, Cergol KM, Salam NK, Bulloch EM, Chi G, Pang A, Britton WJ, West NP, Baker EN, Lott JS, Payne RJ. Synthesis and evaluation of M. tuberculosis salicylate synthase (MbtI) inhibitors designed to probe plasticity in the active site. Org Biomol Chem. 2012 Dec 14;10(46):9223-36. doi: 10.1039/c2ob26736e. Epub 2012, Oct 29. PMID:23108268 doi:http://dx.doi.org/10.1039/c2ob26736e
- ↑ 4.0 4.1 Voss, James J., Kerry Rutter, Benjamin G. Schroedor, Hua Su, and YaQi Zhu. "The salicylate-derived mycobactin siderophores of Mycobacterium tuberculosis are essential for growth in macrophages." Proceedings of the National Academy of Sciences 97.3 (2000): 1252-57. Web. 14 Mar. 2015.
- ↑ Lamb AL. Pericyclic reactions catalyzed by chorismate-utilizing enzymes. Biochemistry. 2011 Sep 6;50(35):7476-83. doi: 10.1021/bi2009739. Epub 2011 Aug, 12. PMID:21823653 doi:http://dx.doi.org/10.1021/bi2009739
- ↑ 6.0 6.1 He Z, Stigers Lavoie KD, Bartlett PA, Toney MD. Conservation of mechanism in three chorismate-utilizing enzymes. J Am Chem Soc. 2004 Mar 3;126(8):2378-85. PMID:14982443 doi:http://dx.doi.org/10.1021/ja0389927
- ↑ Ferrer S, Marti S, Moliner V, Tunon I, Bertran J. Understanding the different activities of highly promiscuous MbtI by computational methods. Phys Chem Chem Phys. 2012 Mar 14;14(10):3482-9. doi: 10.1039/c2cp23149b. Epub, 2012 Feb 3. PMID:22307014 doi:http://dx.doi.org/10.1039/c2cp23149b
- ↑ 8.0 8.1 8.2 8.3 Nicoloff H, Arsene-Ploetze F, Malandain C, Kleerebezem M, Bringel F. Two arginine repressors regulate arginine biosynthesis in Lactobacillus plantarum. J Bacteriol. 2004 Sep;186(18):6059-69. PMID:15342575 doi:http://dx.doi.org/10.1128/JB.186.18.6059-6069.2004
- ↑ Ferrer S, Marti S, Moliner V, Tunon I, Bertran J. Understanding the different activities of highly promiscuous MbtI by computational methods. Phys Chem Chem Phys. 2012 Mar 14;14(10):3482-9. doi: 10.1039/c2cp23149b. Epub, 2012 Feb 3. PMID:22307014 doi:http://dx.doi.org/10.1039/c2cp23149b
- ↑ Ferrer S, Marti S, Moliner V, Tunon I, Bertran J. Understanding the different activities of highly promiscuous MbtI by computational methods. Phys Chem Chem Phys. 2012 Mar 14;14(10):3482-9. doi: 10.1039/c2cp23149b. Epub, 2012 Feb 3. PMID:22307014 doi:http://dx.doi.org/10.1039/c2cp23149b
- ↑ Ferrer S, Marti S, Moliner V, Tunon I, Bertran J. Understanding the different activities of highly promiscuous MbtI by computational methods. Phys Chem Chem Phys. 2012 Mar 14;14(10):3482-9. doi: 10.1039/c2cp23149b. Epub, 2012 Feb 3. PMID:22307014 doi:http://dx.doi.org/10.1039/c2cp23149b
- ↑ Ferrer S, Marti S, Moliner V, Tunon I, Bertran J. Understanding the different activities of highly promiscuous MbtI by computational methods. Phys Chem Chem Phys. 2012 Mar 14;14(10):3482-9. doi: 10.1039/c2cp23149b. Epub, 2012 Feb 3. PMID:22307014 doi:http://dx.doi.org/10.1039/c2cp23149b
- ↑ Tuberculosis (TB). Ed. Sam Posner. Centers for Disease Control and Prevention, n.d. Web. 9 Apr. 2015.
Student contributors
Stephanie Raynor and Robin Gagnon
Related pdb files and proteopedia pages
3D structures of isochorismate pyruvate lyase
3log – MtIPL/isochorismate synthase - Mycobacterium tuberculosis
3rv6, 3rv7, 3rv8, 3rv9, 3st6, 3veh - MtIPL/isochorismate synthase + inhibitor
2h9c – PaIPL residues 1-99 – Pseudomonas aeruginosa
2h9d - PaIPL + pyruvate
3LOG
3D structure of isochorismate synthase
2eua, 3bzm, 3bzn - MenF from E. coli
3os6 - DhbC from Bacillus anthracis
3gse - MenF from Yersinia pestis
3hwo - EntC
3D structure of salicylate synthase
3veh - MbtI with inhibitor methylAMT
3st6 - MbtI with isochorismate analogue inhibitor
3rv6 (Phenyl R-group), 3rv7 (Isopropyl R-group), 3rv8 (Cyclopropyl R-group), 3rv9 (Ethyl R-group) - MbtI with inhibitor
2fn0, 2fn1 (with products salicylate and pyruvate) - Irp9 from Yersinia enterocolitica
2i6y - MbtI