This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
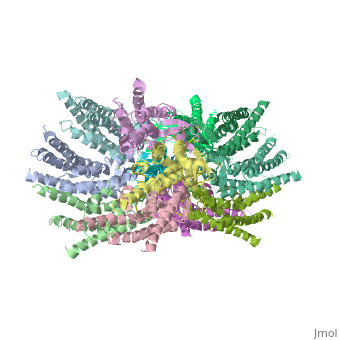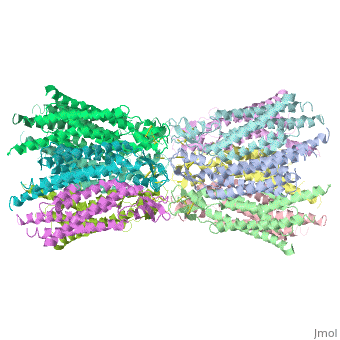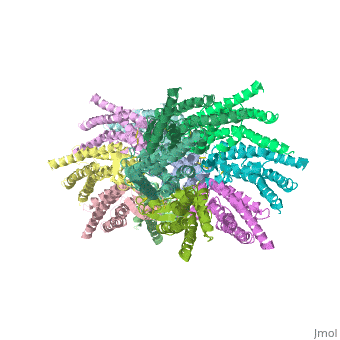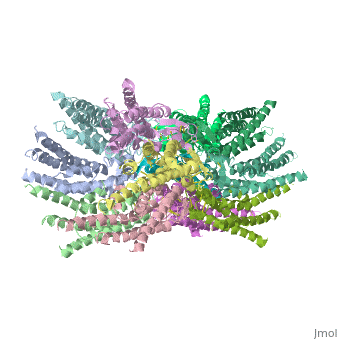
Connexin
From Proteopedia
(Difference between revisions)
| Line 22: | Line 22: | ||
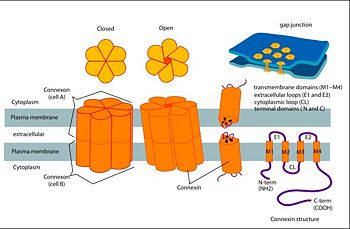
==Structure of the cx26 protomer:== | ==Structure of the cx26 protomer:== | ||
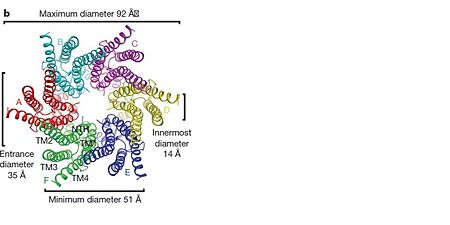
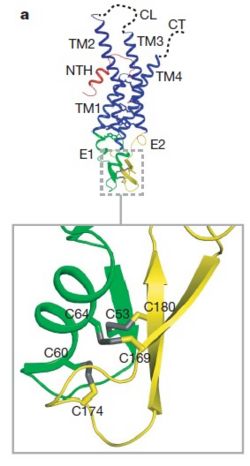
| - | As mentioned before the connexin [http://en.wikipedia.org/wiki/Promoter_(genetics) protomer] has <scene name='70/701426/Connexin_structure/1'>four transmembrane (TM1–4), two extracellular loops , a cytoplasmic loop, an N-terminal helix (NTH), and a C-terminal segment </scene>. Cx26 forms a typical four-helix bundle in which any pair of adjacent helices is antiparallel. The major pore-lining helix TM1 is inclined, so that the pore diameter narrows from the cytoplasmic to the extracellular side of the membrane, and ends in a short 3<sub>10</sub> helix. | + | As mentioned before the connexin [http://en.wikipedia.org/wiki/Promoter_(genetics) protomer] has <scene name='70/701426/Connexin_structure/1'>four transmembrane (TM1–4), two extracellular loops , a cytoplasmic loop, an N-terminal helix (NTH), and a C-terminal segment </scene>. Cx26 forms a typical four-helix bundle in which any pair of adjacent helices is antiparallel. The major pore-lining helix TM1 is inclined, so that the pore diameter narrows from the cytoplasmic to the extracellular side of the membrane, and ends in a short [http://en.wikipedia.org/wiki/310_helix 3<sub>10</sub> helix]. |
| - | The extracellular loop <scene name='70/701426/E1_and_e2/1'>E1</scene> contains a 3<sub>10</sub> helix at the beginning and a short α-helix in its C-terminal. E2, together with E1, contains a short antiparallel β-sheet and stretches over E1, forming the outside <scene name='70/701426/E1_and_e2/1'>wall of the connexon</scene>. Six conserved cysteine residues, three in each loop, form intramolecular disulphide bonds between E1 and E2 Most of the prominent intra-protomer interactions are in the extracellular part of the transmembrane region, The interactions between the two adjoining connexons of the gap junction channel, which involve both E1 and E2 .[[image:E1.jpg | thumb | center | 250px]] | + | The extracellular loop <scene name='70/701426/E1_and_e2/1'>E1</scene> contains a [http://en.wikipedia.org/wiki/310_helix 3<sub>10</sub> helix] at the beginning and a short α-helix in its C-terminal. E2, together with E1, contains a short antiparallel β-sheet and stretches over E1, forming the outside <scene name='70/701426/E1_and_e2/1'>wall of the connexon</scene>. Six conserved cysteine residues, three in each loop, form intramolecular disulphide bonds between E1 and E2 Most of the prominent intra-protomer interactions are in the extracellular part of the transmembrane region, The interactions between the two adjoining connexons of the gap junction channel, which involve both E1 and E2 .[[image:E1.jpg | thumb | center | 250px]] |
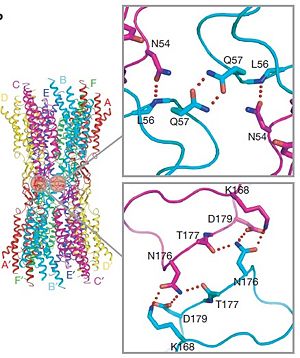
The N-terminal half of E2 seems rather flexible and its amino-acid sequence varies greatly among connexins . The C-terminal half of E2 begins with a 310 turn is followed by a conserved Pro-Cys-Pro motif that reverses its direction back to TM4. Most of the prominent intra-protomer interactions are in the extracellular part of the transmembrane region . Arg 32 (TM1) interactswithGln 80 (TM2),Glu 147 (TM3), and Ser 199 (TM4). Two hydrophobic cores around Trp 44 (E1) and Trp 77 (TM2) stabilize the protomer structure. Ala 39 (TM1), Ala 40 (TM1), Val 43 (E1) and Ile 74 (TM2) contribute to the first hydrophobic core around Trp 44, and Phe 154 (TM3) and Met 195 (TM4) form the second core with Trp 77 . In the intracellular part of the transmembrane region, Arg 143 (TM3) forms hydrogen bonds with Asn 206 (TM3) and Ser 139 (TM3) .<ref name='Structure'/> | The N-terminal half of E2 seems rather flexible and its amino-acid sequence varies greatly among connexins . The C-terminal half of E2 begins with a 310 turn is followed by a conserved Pro-Cys-Pro motif that reverses its direction back to TM4. Most of the prominent intra-protomer interactions are in the extracellular part of the transmembrane region . Arg 32 (TM1) interactswithGln 80 (TM2),Glu 147 (TM3), and Ser 199 (TM4). Two hydrophobic cores around Trp 44 (E1) and Trp 77 (TM2) stabilize the protomer structure. Ala 39 (TM1), Ala 40 (TM1), Val 43 (E1) and Ile 74 (TM2) contribute to the first hydrophobic core around Trp 44, and Phe 154 (TM3) and Met 195 (TM4) form the second core with Trp 77 . In the intracellular part of the transmembrane region, Arg 143 (TM3) forms hydrogen bonds with Asn 206 (TM3) and Ser 139 (TM3) .<ref name='Structure'/> | ||
[[image:bonds.jpg | thumb | center | 300px]] | [[image:bonds.jpg | thumb | center | 300px]] | ||
Revision as of 15:59, 19 May 2015
| |||||||||||
References
- ↑ Zonta F, Buratto D, Cassini C, Bortolozzi M, Mammano F. Molecular dynamics simulations highlight structural and functional alterations in deafness-related M34T mutation of connexin 26. Front Physiol. 2014 Mar 4;5:85. doi: 10.3389/fphys.2014.00085. eCollection 2014. PMID:24624091 doi:http://dx.doi.org/10.3389/fphys.2014.00085
- ↑ 2.0 2.1 2.2 2.3 Suga M, Maeda S, Nakagawa S, Yamashita E, Tsukihara T. A description of the structural determination procedures of a gap junction channel at 3.5 A resolution. Acta Crystallogr D Biol Crystallogr. 2009 Aug;65(Pt 8):758-66. Epub 2009, Jul 10. PMID:19622859 doi:http://dx.doi.org/10.1107/S0907444909014711
- ↑ http://en.wikipedia.org/wiki/Connexin
- ↑ 4.0 4.1 Ambrosi C, Walker AE, Depriest AD, Cone AC, Lu C, Badger J, Skerrett IM, Sosinsky GE. Analysis of trafficking, stability and function of human connexin 26 gap junction channels with deafness-causing mutations in the fourth transmembrane helix. PLoS One. 2013 Aug 15;8(8):e70916. doi: 10.1371/journal.pone.0070916. eCollection, 2013. PMID:23967136 doi:http://dx.doi.org/10.1371/journal.pone.0070916
- ↑ 5.0 5.1 5.2 Oshima A, Tani K, Toloue MM, Hiroaki Y, Smock A, Inukai S, Cone A, Nicholson BJ, Sosinsky GE, Fujiyoshi Y. Asymmetric Configurations and N-terminal Rearrangements in Connexin26 Gap Junction Channels. J Mol Biol. 2011 Jan 21;405(3):724-35. Epub 2010 Nov 20. PMID:21094651 doi:10.1016/j.jmb.2010.10.032
Proteopedia Page Contributors and Editors (what is this?)
Safaa Salah Hussiesy, Michal Harel, Doaa Naffaa, Jaime Prilusky