This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1122
From Proteopedia
(Difference between revisions)
| Line 31: | Line 31: | ||
Bcl-2 localized on external mitochondrial membrane can also inhibit the release of cytochrome c from mitochondria. <ref>[http://science.sciencemag.org/content/275/5303/1132.full The Release of Cytochrome c from Mitochondria: A Primary Site for Bcl-2 Regulation of Apoptosis] </ref> <ref>[http://science.sciencemag.org/content/275/5303/1129.full Prevention of Apoptosis by Bcl-2: Release of Cytochrome c from Mitochondria Blocked] </ref> | Bcl-2 localized on external mitochondrial membrane can also inhibit the release of cytochrome c from mitochondria. <ref>[http://science.sciencemag.org/content/275/5303/1132.full The Release of Cytochrome c from Mitochondria: A Primary Site for Bcl-2 Regulation of Apoptosis] </ref> <ref>[http://science.sciencemag.org/content/275/5303/1129.full Prevention of Apoptosis by Bcl-2: Release of Cytochrome c from Mitochondria Blocked] </ref> | ||
| + | |||
| + | === Regulation of proinflammatory caspase-1 activation === | ||
| + | |||
| + | NALP1 is a member of a NLR-family proteins. Its function is to activate the members of the proinflammatory caspase family which participate in cytokines activation pathway (especially caspase-1). The Bcl-2 loop regions between the BH3 and BH4 bind NALP1. This interaction is exclusively reserved to two members of Bcl-2 family: Bcl-XL and Bcl-2 itself because this interacting region is highly variable in Bcl-2 family. By binding to NALP1, Bcl-2 inhibits the inflammatory caspase activation. Hence, it protects cell from the stress. | ||
| + | The posttranslational modifications found on the loops between BH3 and BH4 modify the anti-apoptotic activity of Bcl-2. Hence, the Bcl-2 binding to NALP1 can be affected by these modifications. <ref>[http://www.sciencedirect.com/science/article/pii/S0092867407003042 Bcl-2 and Bcl-XL Regulate Proinflammatory Caspase-1 Activation by Interaction with NALP1] </ref> | ||
== Disease == | == Disease == | ||
Revision as of 16:23, 28 January 2016
| This Sandbox is Reserved from 15/12/2015, through 15/06/2016 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1120 through Sandbox Reserved 1159. |
To get started:
More help: Help:Editing |
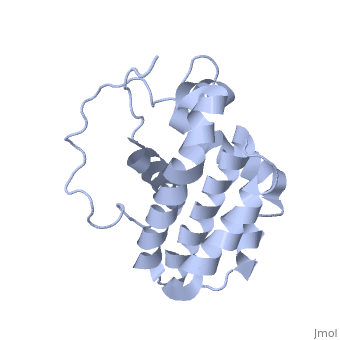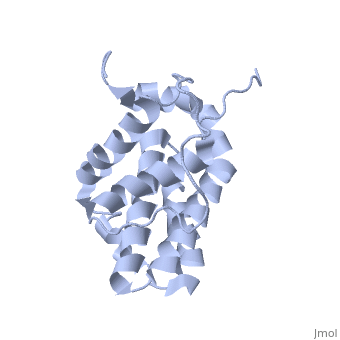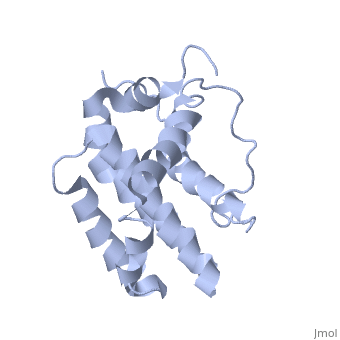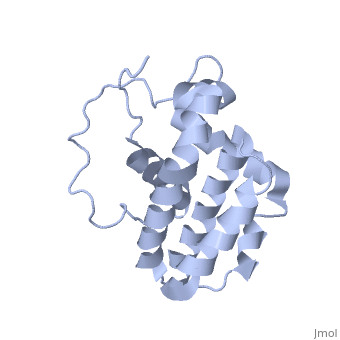
HUMAN BCL-2, ISOFORM1
| |||||||||||
References
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644
- ↑ Solution structure of the antiapoptotic protein bcl-2
- ↑ Alpha-Helical Destabilization of the Bcl-2-BH4-Domain Peptide Abolishes Its Ability to Inhibit the IP3 Receptor
- ↑ Control of mitochondrial apoptosis by the Bcl-2 family
- ↑ Differential Targeting of Prosurvival Bcl-2 Proteins by Their BH3-Only Ligands Allows Complementary Apoptotic Function
- ↑ Distinct BH3 domains either sensitize or activate mitochondrial apoptosis, serving as prototype cancer therapeutics
- ↑ The Release of Cytochrome c from Mitochondria: A Primary Site for Bcl-2 Regulation of Apoptosis
- ↑ Prevention of Apoptosis by Bcl-2: Release of Cytochrome c from Mitochondria Blocked
- ↑ Bcl-2 and Bcl-XL Regulate Proinflammatory Caspase-1 Activation by Interaction with NALP1