This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1122
From Proteopedia
(Difference between revisions)
| Line 2: | Line 2: | ||
==HUMAN BCL-2, ISOFORM1== | ==HUMAN BCL-2, ISOFORM1== | ||
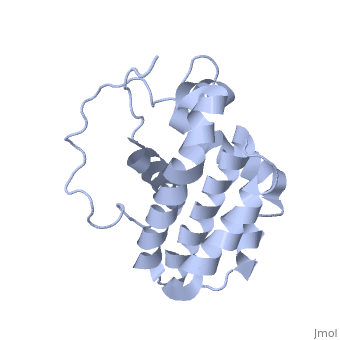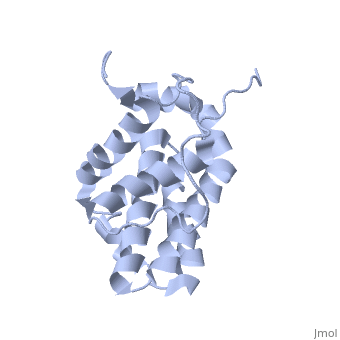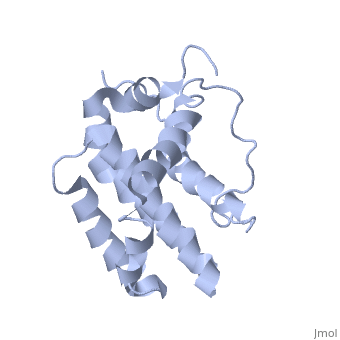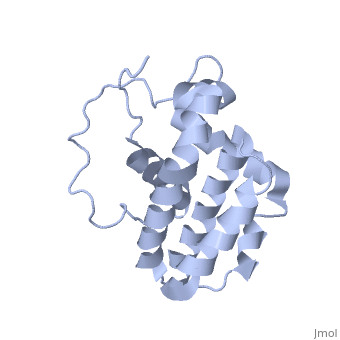
<StructureSection load='1g5m' size='400' side='right' caption='3D STRUCTURE OF HUMAN BCL-2, ISOFORM1 (from residue 3 to 207) BASED ON NMR SPECTROSCOPY' scene=''> | <StructureSection load='1g5m' size='400' side='right' caption='3D STRUCTURE OF HUMAN BCL-2, ISOFORM1 (from residue 3 to 207) BASED ON NMR SPECTROSCOPY' scene=''> | ||
| - | Human | + | Human Bcl-2 (B-Cell Lymphoma 2), isoform 1 is an oncoprotein of 239 residues regulating cell death (apoptosis), notably acting as an anti-apoptotic. It is encoded by the ''BCL-2'' gene located on the 18th chromosome (63.12-63.32 Mb). There are 2 isoforms of this protein (𝛼 and 𝛽), produced by alternative splicing, and which differ by 2 aminoacids (residues 96 (A→T) and 110 (R→G))<ref>[http://www.ncbi.nlm.nih.gov/pmc/articles/PMC30598/ Solution structure of the antiapoptotic protein bcl-2]</ref>. Alteration of this protein is a caused of many cancers, and is also likely to be involved in schyzophrenia and autoimmunity. |
== Structure == | == Structure == | ||
| - | Human | + | Human Bcl-2, isoform 1 is a 26kDa protein of 239 residues which is negatively charged at pH 7. The linear structure highlights 5 domains: <scene name='71/719863/Scenelucas/1'>BH4</scene> (10-30), |
<scene name='71/719863/Scenebh3/2'>BH3</scene> (93-107), <scene name='71/719863/Scenebh1/1'>BH1</scene> (136-155), <scene name='71/719863/Scenebh2/1'>BH2</scene> (187-202) and a | <scene name='71/719863/Scenebh3/2'>BH3</scene> (93-107), <scene name='71/719863/Scenebh1/1'>BH1</scene> (136-155), <scene name='71/719863/Scenebh2/1'>BH2</scene> (187-202) and a | ||
| - | transmembrane domain (212-233) (due to its poor behavior in solution, it has been replaced by a segment of Bcl-xl in the presented 3D structure). It organizes as eight alpha-helices: from 11 to 25 (1) , from 93 to 107 (2), from 109 to 118 (3), from 126 to 137 (4), from 144-163 (5), from 169 to 184 (6), from 186 to 191 (7) and from 194 to 202(8). Helices 5 and 6 are mostly hydrophobic and they are surrounded by four other helices characterized by their amphipathic properties. There are also 3 turns (32-34, 123-125, 138-140). The <scene name='71/719863/Bcl2helix/1'>3rd alpha-helix</scene> is a 3(10) helix, whereas | + | transmembrane domain (212-233) (due to its poor behavior in solution, it has been replaced by a segment of Bcl-xl in the presented 3D structure). It organizes as eight alpha-helices: from 11 to 25 (1) , from 93 to 107 (2), from 109 to 118 (3), from 126 to 137 (4), from 144-163 (5), from 169 to 184 (6), from 186 to 191 (7) and from 194 to 202(8). Helices 5 and 6 are mostly hydrophobic and they are surrounded by four other helices characterized by their amphipathic properties. There are also 3 turns (32-34, 123-125, 138-140). The <scene name='71/719863/Bcl2helix/1'>3rd alpha-helix</scene> is a 3(10) helix, whereas Bcl-Xl 3rd helix is a normal alpha-helix. |
The transmembrane domain of Bcl-2 is made of 21 aminoacids and is located at the carboxy-terminal tail of the protein. It allows the docking of Bcl-2 in the mitochondrial outer membrane where the protein interacts with other effectors.<ref>[http://www.ncbi.nlm.nih.gov/pubmed/24905660 Peptides derived from the transmembrane domain of Bcl-2 proteins as potential mitochondrial priming tools.]</ref> | The transmembrane domain of Bcl-2 is made of 21 aminoacids and is located at the carboxy-terminal tail of the protein. It allows the docking of Bcl-2 in the mitochondrial outer membrane where the protein interacts with other effectors.<ref>[http://www.ncbi.nlm.nih.gov/pubmed/24905660 Peptides derived from the transmembrane domain of Bcl-2 proteins as potential mitochondrial priming tools.]</ref> | ||
| Line 38: | Line 38: | ||
=== Cancer === | === Cancer === | ||
| - | + | Bcl-2 is involved in many cancers such as breast, melanoma, prostate, chronic lymphocytic leukemia and lung cancers. In fact, its overexpression combined with an overexpression of c-Myc (another oncoprotein) produce aggressive B-lymphocytes. <ref>[http://www.bloodjournal.org/content/125/4/658 BCL2 mutations are associated with increased risk of transformation and shortened survival in follicular lymphoma]</ref>. It has also be shown that Bcl-2 overpexpression supresses DNA repair by enhancing Myc transcriptional activity. | |
| - | <ref>[http://www.jbc.org/content/281/20/14446.full | + | <ref>[http://www.jbc.org/content/281/20/14446.full Bcl-2 Suppresses DNA Repair by Enhancing c-Myc Transcriptional Activity]</ref> In fact, Bcl-2 is encoded by the ''BCL-2'' gene (0,20 Mb) located on the 18th chromosome. Nevertheless, translocation between chromosome 14 and 18 juxtaposes the ''BCL-2'' gene and the immunoglobulin locus. This brings the ''BCL-2'' gene under the regulation of the immunoglobulin heavy-chain enhancer, diregulating ''BCL-2'' expression level (involved in non-Hodgkin's lymphomas). <ref>[http://www.nature.com/onc/journal/v27/n50/full/onc2008307a.html Bcl-2 family proteins and cancer]</ref> |
| - | Other mechanisms appear to regulate and enhance the level of expression of BCL-2 : loss of endogenous microRNAs which normally repress | + | Other mechanisms appear to regulate and enhance the level of expression of BCL-2 : loss of endogenous microRNAs which normally repress Bcl-2 expression, and also hypomethylation.<ref>[http://www.nature.com/onc/journal/v27/n50/full/onc2008307a.html Bcl-2 family proteins and cancer]</ref> |
| - | + | Bcl-2 <scene name='71/719863/Scenelucas/1'>BH4</scene> domain mediates the interaction with MBII domain of Myc. This interaction enhance c-Myc half-life, resulting in an enhancing of its total activity. c-Myc is a well known oncoprotein (transcription factor) involved in many cancers as it regulates a lot of genes of the cell cycle. | |
Revision as of 17:31, 28 January 2016
| This Sandbox is Reserved from 15/12/2015, through 15/06/2016 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1120 through Sandbox Reserved 1159. |
To get started:
More help: Help:Editing |
HUMAN BCL-2, ISOFORM1
| |||||||||||
References
- ↑ Solution structure of the antiapoptotic protein bcl-2
- ↑ Peptides derived from the transmembrane domain of Bcl-2 proteins as potential mitochondrial priming tools.
- ↑ Solution structure of the antiapoptotic protein bcl-2
- ↑ Alpha-Helical Destabilization of the Bcl-2-BH4-Domain Peptide Abolishes Its Ability to Inhibit the IP3 Receptor
- ↑ BH1 and BH2 domains of Bcl-2 are required for inhibition of apoptosis and heterodimerization with Bax
- ↑ Control of mitochondrial apoptosis by the Bcl-2 family
- ↑ Differential Targeting of Prosurvival Bcl-2 Proteins by Their BH3-Only Ligands Allows Complementary Apoptotic Function
- ↑ Distinct BH3 domains either sensitize or activate mitochondrial apoptosis, serving as prototype cancer therapeutics
- ↑ The Release of Cytochrome c from Mitochondria: A Primary Site for Bcl-2 Regulation of Apoptosis
- ↑ Prevention of Apoptosis by Bcl-2: Release of Cytochrome c from Mitochondria Blocked
- ↑ Bcl-2 and Bcl-XL Regulate Proinflammatory Caspase-1 Activation by Interaction with NALP1
- ↑ BCL2 mutations are associated with increased risk of transformation and shortened survival in follicular lymphoma
- ↑ Bcl-2 Suppresses DNA Repair by Enhancing c-Myc Transcriptional Activity
- ↑ Bcl-2 family proteins and cancer
- ↑ Bcl-2 family proteins and cancer