This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1126
From Proteopedia
(Difference between revisions)
| Line 21: | Line 21: | ||
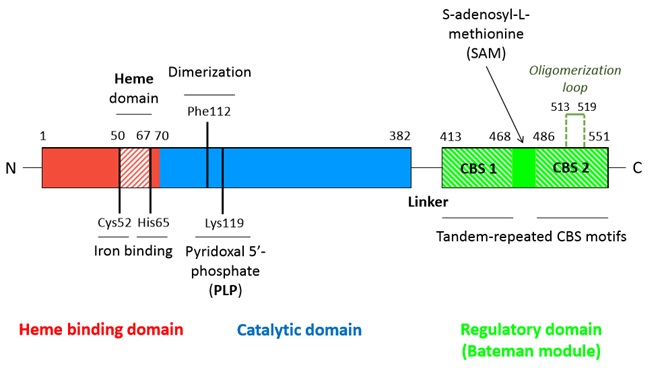
==== Cofactors ==== | ==== Cofactors ==== | ||
| - | *<scene name='71/719867/Scene_4/2'>Heme iron</scene> | + | *<scene name='71/719867/Scene_4/2'>Heme iron</scene> |
The heme is one of the two cofactors of hCBS. | The heme is one of the two cofactors of hCBS. | ||
It is bound in an hydrophobic pocket composed of the residues 50-67. The iron atom is hexacoordinated with the sulfhydryl group of Cys52 and the Nε2 atom of His65 (axial coordination) and with the four nitrogen atoms of the heme. | It is bound in an hydrophobic pocket composed of the residues 50-67. The iron atom is hexacoordinated with the sulfhydryl group of Cys52 and the Nε2 atom of His65 (axial coordination) and with the four nitrogen atoms of the heme. | ||
| Line 27: | Line 27: | ||
It is supposed to act as a redox sensor or as a way to facilitate a correct folding. | It is supposed to act as a redox sensor or as a way to facilitate a correct folding. | ||
| - | *<scene name='71/719867/Scene_4/1'>Pyridoxal phosphate (PLP)</scene> | + | *<scene name='71/719867/Scene_4/1'>Pyridoxal phosphate (PLP)</scene> |
Revision as of 19:21, 29 January 2016
| This Sandbox is Reserved from 15/12/2015, through 15/06/2016 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1120 through Sandbox Reserved 1159. |
To get started:
More help: Help:Editing |
Human cystathionine β-synthase (hCBS)
| |||||||||||