This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Daniel Schemenauer/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 18: | Line 18: | ||
===Interactions with a Negative Allosteric Modulator=== | ===Interactions with a Negative Allosteric Modulator=== | ||
The structure of mGlu<sub>5</sub> bound to the NAM [https://en.wikipedia.org/wiki/Mavoglurant Mavoglurant] demonstrates how protein activity is decreased through drug interactions. | The structure of mGlu<sub>5</sub> bound to the NAM [https://en.wikipedia.org/wiki/Mavoglurant Mavoglurant] demonstrates how protein activity is decreased through drug interactions. | ||
| - | <scene name='72/726404/Scene_7/2'>Mavoglurant</scene> binds within the allosteric binding site in the core of the seven trans-membrane α-helices,having passed through the restricted entrance formed by the ECL2. Bound Mavoglurant forms multiple interactions with the protein that further stabilize the inactive conformation. | + | <scene name='72/726404/Scene_7/2'>Mavoglurant</scene> binds within the allosteric binding site in the core of the seven trans-membrane α-helices,having passed through the restricted entrance formed by the <scene name='72/726409/Mavoglurant_overview2/3'>ECL2</scene>. Bound Mavoglurant forms multiple interactions with the protein that further stabilize the inactive conformation. |
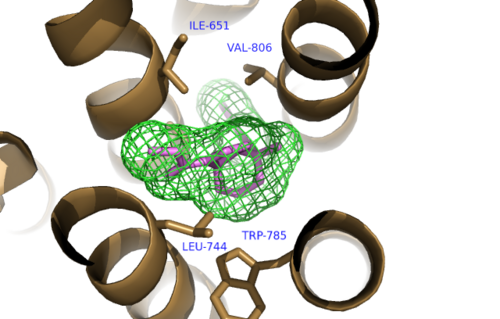
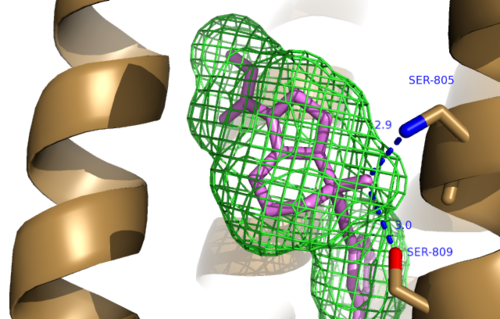
The bicyclic ring system of the drug is surrounded by a pocket of mainly hydrophobic residues including Val 806, Met 802, Phe 788, Trp 785, Leu 744, Ile 651, Pro 655 and Asn 747<ref name="Primary">PMID: 25042998 </ref> (Figure 1). The carbamate tail of mavoglurant forms a hydrogen bond through its carbonyl oxygen to the amide side-chain of Asparagine 747 of TM4 (Figure 2). A hydroxyl group similarly forms hydrogen bonds to the protein, specifically to two serine residues (S805 and S809) of TM7 which form a hydrogen bonding network to other residues through their main chain atoms and a coordinated water molecule (omitted for clarity) (Figure 3). The interactions between Mavoglurant andmGlu<sub>5</sub> involved TM helices that were not previously stabilized by any strong interactions, introducing a new level of stability that favors the inactive conformation of the protein and hence decrease activity<ref name="Primary">PMID: 25042998 </ref>. | The bicyclic ring system of the drug is surrounded by a pocket of mainly hydrophobic residues including Val 806, Met 802, Phe 788, Trp 785, Leu 744, Ile 651, Pro 655 and Asn 747<ref name="Primary">PMID: 25042998 </ref> (Figure 1). The carbamate tail of mavoglurant forms a hydrogen bond through its carbonyl oxygen to the amide side-chain of Asparagine 747 of TM4 (Figure 2). A hydroxyl group similarly forms hydrogen bonds to the protein, specifically to two serine residues (S805 and S809) of TM7 which form a hydrogen bonding network to other residues through their main chain atoms and a coordinated water molecule (omitted for clarity) (Figure 3). The interactions between Mavoglurant andmGlu<sub>5</sub> involved TM helices that were not previously stabilized by any strong interactions, introducing a new level of stability that favors the inactive conformation of the protein and hence decrease activity<ref name="Primary">PMID: 25042998 </ref>. | ||
[[Image:Mav_Hydrophobic_pocket.png |500 px|left|thumb|Figure 1.Hydrophobic Pocket Surrounding Mavoglurant]] | [[Image:Mav_Hydrophobic_pocket.png |500 px|left|thumb|Figure 1.Hydrophobic Pocket Surrounding Mavoglurant]] | ||
Revision as of 12:11, 29 March 2016
| |||||||||||
References
- ↑ Vassilatis DK, Hohmann JG, Zeng H, Li F, Ranchalis JE, Mortrud MT, Brown A, Rodriguez SS, Weller JR, Wright AC, Bergmann JE, Gaitanaris GA. The G protein-coupled receptor repertoires of human and mouse. Proc Natl Acad Sci U S A. 2003 Apr 15;100(8):4903-8. Epub 2003 Apr 4. PMID:12679517 doi:http://dx.doi.org/10.1073/pnas.0230374100
- ↑ 2.0 2.1 Venkatakrishnan AJ, Deupi X, Lebon G, Tate CG, Schertler GF, Babu MM. Molecular signatures of G-protein-coupled receptors. Nature. 2013 Feb 14;494(7436):185-94. doi: 10.1038/nature11896. PMID:23407534 doi:http://dx.doi.org/10.1038/nature11896
- ↑ Pin JP, Galvez T, Prezeau L. Evolution, structure, and activation mechanism of family 3/C G-protein-coupled receptors. Pharmacol Ther. 2003 Jun;98(3):325-54. PMID:12782243
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 4.9 Dore AS, Okrasa K, Patel JC, Serrano-Vega M, Bennett K, Cooke RM, Errey JC, Jazayeri A, Khan S, Tehan B, Weir M, Wiggin GR, Marshall FH. Structure of class C GPCR metabotropic glutamate receptor 5 transmembrane domain. Nature. 2014 Jul 31;511(7511):557-62. doi: 10.1038/nature13396. Epub 2014 Jul 6. PMID:25042998 doi:http://dx.doi.org/10.1038/nature13396
- ↑ Shigemoto R, Nomura S, Ohishi H, Sugihara H, Nakanishi S, Mizuno N. Immunohistochemical localization of a metabotropic glutamate receptor, mGluR5, in the rat brain. Neurosci Lett. 1993 Nov 26;163(1):53-7. PMID:8295733
- ↑ Li G, Jorgensen M, Campbell BM. Metabotropic glutamate receptor 5-negative allosteric modulators for the treatment of psychiatric and neurological disorders (2009-July 2013). Pharm Pat Anal. 2013 Nov;2(6):767-802. doi: 10.4155/ppa.13.58. PMID:24237242 doi:http://dx.doi.org/10.4155/ppa.13.58