This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1160
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
== Human metabotropic glutamate receptor 5 transmembrane domain == | == Human metabotropic glutamate receptor 5 transmembrane domain == | ||
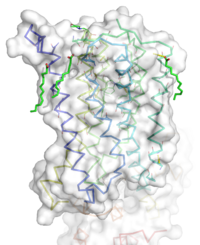
<StructureSection load='4oo9' size='300' frame='true' side='right' caption='Human metabotropic glutamate receptor 5 transmembrane domain' scene='72/721531/Protien_clean_sce/1'> | <StructureSection load='4oo9' size='300' frame='true' side='right' caption='Human metabotropic glutamate receptor 5 transmembrane domain' scene='72/721531/Protien_clean_sce/1'> | ||
| - | Receiving and responding to extracellular messages is critical to the proper function of the nervous system. Glutamate is the major excitory neurotransmitter of the CNS, and metabotropic glutamate receptor 5 will play a major role in glutamate signaling. Metabotropic glutamate receptor 5 transmembrane domain is a homodimeric [[GPCR]] that resides in the cellular membrane <ref name="Dore" />. This domain is a member of the Class C GPCR family and can further be categorized into the Group I subgroup<ref name="Wu" />. The transmembrane domain will signal through a Gq/11 pathway<ref name="Dore" />. mGlu5 will bind glutamate to the extracellular Venus flytrap domain and the signal will be transduced across the membrane to a heterotrimeric G protein, which will ultimately lead to calcium release and activation of PKC<ref name="Wu" />. This will elicit a excitory post-synaptic repose and modulate long term potentiation<ref name="Wu" />. Human metabotropic glutamate receptor 5 is found throughout the central nervous system. Areas containing high concentrations of this protein are often involved in emotional processing and higher cognition<ref name="Niswender" />. The localization of mGlu5 in the CNS and the presence of multiple domains makes mGlu5 a possible target for treating schizophrenia,[http://www.fragilex.org/fragile-x/fragile-x-syndrome/ Fragile X], depression, anxiety, and Alzheimer's disease<ref name="Wu" />. | + | Receiving and responding to extracellular messages is critical to the proper function of the nervous system. Glutamate is the major excitory neurotransmitter of the CNS, and metabotropic glutamate receptor 5 will play a major role in glutamate signaling. Metabotropic glutamate receptor 5 transmembrane domain is a homodimeric [[GPCR]] that resides in the cellular membrane <ref name="Dore" />. This domain is a member of the Class C GPCR family and can further be categorized into the Group I subgroup<ref name="Wu" />. The transmembrane domain will signal through a Gq/11 pathway<ref name="Dore" />. mGlu5 will bind glutamate to the extracellular Venus flytrap domain and the signal will be transduced across the membrane to a heterotrimeric G protein, which will ultimately lead to calcium release and activation of [[PKC]]<ref name="Wu" />. This will elicit a excitory post-synaptic repose and modulate long term potentiation<ref name="Wu" />. Human metabotropic glutamate receptor 5 is found throughout the central nervous system. Areas containing high concentrations of this protein are often involved in emotional processing and higher cognition<ref name="Niswender" />. The localization of mGlu5 in the CNS and the presence of multiple domains makes mGlu5 a possible target for treating schizophrenia,[http://www.fragilex.org/fragile-x/fragile-x-syndrome/ Fragile X], depression, anxiety, and Alzheimer's disease<ref name="Wu" />. |
== Discovery == | == Discovery == | ||
| Line 24: | Line 24: | ||
=== Ionic Locks === | === Ionic Locks === | ||
| - | Another important structural feature of the protein is the series of <scene name='72/721531/Ionic_lock/2'>ionic locks</scene> on the intracellular side of the protein. Interaction between amino acids will form a salt bridge which will stabilize the inactive conformation<ref name="Dore" />. The primary ionic lock forms between Glu770, Lys665, and Ser613<ref name="Dore" />. A secondary ionic lock occurs between Ser614 and Arg668<ref name="Dore" />. The purpose of these ionic locks is analogous to the ionic interactions that stabilize the T state in [[Hemoglobin]]. In the case of the TMD, when the NAM mavoglurant is bound the ionic lock is formed. This stabilizes the inactive state, where the intracellular loops are stabilized inwards<ref name="Wu" />. This will effectively block the crevice that is involved in binding the G-protein<ref name="Wu" />. Models have suggested that, even in a glutamate bound state, the mavoglurant bound receptor would be dimerized but incapable of signaling<ref name="Wu" />. This helps maintain the readiness of the pathway, while still decreasing signal response. | + | Another important structural feature of the protein is the series of <scene name='72/721531/Ionic_lock/2'>ionic locks</scene> on the intracellular side of the protein. Interaction between amino acids will form a salt bridge, which will stabilize the inactive conformation<ref name="Dore" />. The primary ionic lock forms between Glu770, Lys665, and Ser613<ref name="Dore" />. A secondary ionic lock occurs between Ser614 and Arg668<ref name="Dore" />. The purpose of these ionic locks is analogous to the ionic interactions that stabilize the T state in [[Hemoglobin]]. In the case of the TMD, when the NAM mavoglurant is bound the ionic lock is formed. This stabilizes the inactive state, where the intracellular loops are stabilized inwards<ref name="Wu" />. This will effectively block the crevice that is involved in binding the G-protein<ref name="Wu" />. Models have suggested that, even in a glutamate bound state, the mavoglurant bound receptor would be dimerized but incapable of signaling<ref name="Wu" />. This helps maintain the readiness of the pathway, while still decreasing signal response. |
== Function and Pathway == | == Function and Pathway == | ||
| - | It all begins with glutamate binding to the venus fly trap domain. The signal transduction goes across the cystine-rich domain to the TMD. Next the dimerization of the TMD occurs. This activates the Gq/11 pathway, which activates phspholipase Cβ<ref name="Niswender" />. The active phospholipase Cβ performs hydrolysis on phosphotinositides and generates inositol 1,4,5-trisphosphate and diacyl-glycerol<ref name="Woodcock" />. This results in calcium mobilization and activation of | + | It all begins with glutamate binding to the venus fly trap domain. The signal transduction goes across the cystine-rich domain to the TMD. Next the dimerization of the TMD occurs. This activates the Gq/11 pathway, which activates phspholipase Cβ<ref name="Niswender" />. The active phospholipase Cβ performs hydrolysis on phosphotinositides and generates inositol 1,4,5-trisphosphate and diacyl-glycerol<ref name="Woodcock" />. This results in calcium mobilization and activation of PKC<ref name="Niswender" />. |
== Disease == | == Disease == | ||
| Line 44: | Line 44: | ||
== External Resources == | == External Resources == | ||
[http://www.fraxa.org/novartis-discontinues-development-mavoglurant-afq056-fragile-x-syndrome/ Novartis Fragile X trials] | [http://www.fraxa.org/novartis-discontinues-development-mavoglurant-afq056-fragile-x-syndrome/ Novartis Fragile X trials] | ||
| + | [http://www.en.wikipedia.org/wiki/Gq_alpha_subunit] | ||
Revision as of 11:26, 30 March 2016
Human metabotropic glutamate receptor 5 transmembrane domain
| |||||||||||