This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1176
From Proteopedia
(Difference between revisions)
| Line 7: | Line 7: | ||
==Introduction== | ==Introduction== | ||
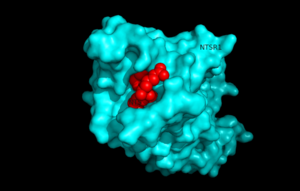
[[Image:surfaceprotein.png |300 px|below|thumb|Figure 1.Top view of NTSR1 protein interacting with NTS ligand]] | [[Image:surfaceprotein.png |300 px|below|thumb|Figure 1.Top view of NTSR1 protein interacting with NTS ligand]] | ||
| - | Neurotensin receptor 1 (NTSR1) is a G-protein coupled receptor (GPCR) that binds to the 13 amino acid peptide, neurotensin. Studies determining the structure of NTSR1 crystallized the GPCR bound with the C-terminus of its tridecapeptide ligand, <scene name='72/721548/Neurotensin/3'>NTS(8-13)</scene> because it has a higher potency and efficacy than its full-length counterpart. NTSR1 is a class A GPCR, and like all G-proteins, consists 7 transmembrane helices. Class A GPCRs bind their ligands within the transmembrane core, and this is seen in NTSR1. Along with the ligand binding pocket at the top of the protein, NTSR1 also contains an allosteric Na+ ion binding pocket underneath. NTS binds to NTSR1, leading to a conformational change of the protein and modulation of second messengers. NTS has been shown to have a variety of biological activities including a role in the leptin signalling pathways, tumor growth, and dopamine regulation. The majority of effects of NTS are mediated through NTSR1. Research of the structure of NTSR1 has focused on the differences between its active and active-like states. | + | Neurotensin receptor 1 (NTSR1) is a G-protein coupled receptor (GPCR) that binds to the 13 amino acid peptide, neurotensin<ref name="SONT">PMID:23051748</ref>. Studies determining the structure of NTSR1 crystallized the GPCR bound with the C-terminus of its tridecapeptide ligand, <scene name='72/721548/Neurotensin/3'>NTS(8-13)</scene> because it has a higher potency and efficacy than its full-length counterpart<ref name="SONT"/>. NTSR1 is a class A GPCR, and like all G-proteins, consists 7 transmembrane helices. Class A GPCRs bind their ligands within the transmembrane core, and this is seen in NTSR1. Along with the ligand binding pocket at the top of the protein, NTSR1 also contains an allosteric Na+ ion binding pocket underneath. NTS binds to NTSR1, leading to a conformational change of the protein and modulation of second messengers. NTS has been shown to have a variety of biological activities including a role in the leptin signalling pathways, tumor growth, and dopamine regulation. The majority of effects of NTS are mediated through NTSR1. Research of the structure of NTSR1 has focused on the differences between its active and active-like states. |
== Structure == | == Structure == | ||
Revision as of 18:08, 1 April 2016
| This Sandbox is Reserved from Jan 11 through August 12, 2016 for use in the course CH462 Central Metabolism taught by R. Jeremy Johnson at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1160 through Sandbox Reserved 1184. |
To get started:
More help: Help:Editing |
Rattus norevegicus NTSR1
| |||||||||||
References
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644
- ↑ White JF, Noinaj N, Shibata Y, Love J, Kloss B, Xu F, Gvozdenovic-Jeremic J, Shah P, Shiloach J, Tate CG, Grisshammer R. Structure of the agonist-bound neurotensin receptor. Nature. 2012 Oct 25;490(7421):508-13. doi: 10.1038/nature11558. Epub 2012 Oct 10. PMID:23051748 doi:http://dx.doi.org/10.1038/nature11558
- ↑ Krumm BE, White JF, Shah P, Grisshammer R. Structural prerequisites for G-protein activation by the neurotensin receptor. Nat Commun. 2015 Jul 24;6:7895. doi: 10.1038/ncomms8895. PMID:26205105 doi:http://dx.doi.org/10.1038/ncomms8895
- ↑ Carraway RE, Plona AM. Involvement of neurotensin in cancer growth: evidence, mechanisms and development of diagnostic tools. Peptides. 2006 Oct;27(10):2445-60. Epub 2006 Aug 2. PMID:16887236 doi:http://dx.doi.org/10.1016/j.peptides.2006.04.030
- ↑ Griebel G, Holsboer F. Neuropeptide receptor ligands as drugs for psychiatric diseases: the end of the beginning? Nat Rev Drug Discov. 2012 May 18;11(6):462-78. doi: 10.1038/nrd3702. PMID:22596253 doi:http://dx.doi.org/10.1038/nrd3702
- ↑ Liang Y, Boules M, Li Z, Williams K, Miura T, Oliveros A, Richelson E. Hyperactivity of the dopaminergic system in NTS1 and NTS2 null mice. Neuropharmacology. 2010 Jun;58(8):1199-205. doi:, 10.1016/j.neuropharm.2010.02.015. Epub 2010 Mar 6. PMID:20211191 doi:http://dx.doi.org/10.1016/j.neuropharm.2010.02.015
- ↑ 8.0 8.1 White JF, Noinaj N, Shibata Y, Love J, Kloss B, Xu F, Gvozdenovic-Jeremic J, Shah P, Shiloach J, Tate CG, Grisshammer R. Structure of the agonist-bound neurotensin receptor. Nature. 2012 Oct 25;490(7421):508-13. doi: 10.1038/nature11558. Epub 2012 Oct 10. PMID:23051748 doi:http://dx.doi.org/10.1038/nature11558