This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1180
From Proteopedia
| Line 13: | Line 13: | ||
==Structural Considerations== | ==Structural Considerations== | ||
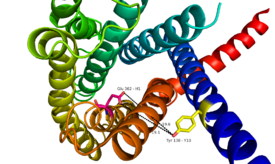
The class B GPCRs, of which GCGR is a member, are different from other Class A GPCRs in several ways. The first is that class B GPCRs contain a protrusion known as a 'stalk,' which is a three α-helical turn elongation of the N-terminus that protrudes past the extracellular (EC) membrane. Structural integrity of this domain in GCGR is essential to ligand binding affinity. (Fig's 1 and 2) | The class B GPCRs, of which GCGR is a member, are different from other Class A GPCRs in several ways. The first is that class B GPCRs contain a protrusion known as a 'stalk,' which is a three α-helical turn elongation of the N-terminus that protrudes past the extracellular (EC) membrane. Structural integrity of this domain in GCGR is essential to ligand binding affinity. (Fig's 1 and 2) | ||
| + | |||
| + | <scene name='72/721552/Ligand_binding_interactions/1'>Ligand Binding Interactions and Crucial Disulfide Bond</scene> | ||
Secondly, the extracellular loop 1 (ECL1) is 3-4 times longer than comparable loops in class A GPCRs, and also affects ligand binding affinity. (Fig. 3)<ref name= "Siu 2013"/> | Secondly, the extracellular loop 1 (ECL1) is 3-4 times longer than comparable loops in class A GPCRs, and also affects ligand binding affinity. (Fig. 3)<ref name= "Siu 2013"/> | ||
Revision as of 22:01, 20 April 2016
G protein coupled receptors (GPCRs) are recognized as the largest known class of integral membrane proteins and are divided into five families; the rhodopsin family (class A), the secretin family (class B), the adhesion family, the glutamate family (class C), and the frizzled/taste family (class F). Roughly 5% of the human genome encodes g-protein-coupled receptors which are responsible for the transduction of endogenous signals and the instigation of cellular response. The variants all contain a similar seven α-helical transmembrane domain (TMD or 7TMD) that, once bound to its peptide ligand, undergoes conformational change and tranduces a signal to coupled, heterotrimeric G proteins which initiate intracellular signal pathways and generate physiological and pathological processes. [1] Class B GPCRs contain 15 distinct receptors for peptide hormones and generate their signal pathway through the activation of adenylate cyclase (AC) which increases concentration of cAMP, inositol phosphate, and calcium levels in cyto. [2] These signals are essential elements of intracellular signal cascades for human diseases including type II diabetes mellitus, osteoporosis, obesity, cancer, neurological degeneration, cardiovascular diseases, headaches, and psychiatric disorders; making their regulation through drug targeting of particular interest to companies developing novel molecules. [3] Structurally based approaches to the development of small-molecule agonists and antagonists have been hampered by the lack of accurate Class B TMD visualizations until recent crystal structures of corticoptropin-releasing factor receptor 1 and human glucagon were realized. [4] [5]
The glucagon class B GPCR (GCGR) is involved in glucose homeostasis through the binding of the signal peptide glucagon. Glucagon ReceptorGlucagon is released from pancreatic α-cells when blood glucose levels fall after a period of fasting or several hours following intake of dietary carbohydrates. Once the peptide hormone is released, it binds to GCGR which is a 485 amino acid protein found in the liver, kidney, intestinal smooth muscle, brain, and adipose tissues. [6] Upon binding, signaling is initiated to heterotrimeric G-proteins containing Gαs. [7] Additionally, GCGR can regulate additional signal pathways including G-proteins of the Gαi family through the adoption of differing receptor conformations. [8] Structural ConsiderationsThe class B GPCRs, of which GCGR is a member, are different from other Class A GPCRs in several ways. The first is that class B GPCRs contain a protrusion known as a 'stalk,' which is a three α-helical turn elongation of the N-terminus that protrudes past the extracellular (EC) membrane. Structural integrity of this domain in GCGR is essential to ligand binding affinity. (Fig's 1 and 2)
Secondly, the extracellular loop 1 (ECL1) is 3-4 times longer than comparable loops in class A GPCRs, and also affects ligand binding affinity. (Fig. 3)[5] Most notably, class B GPCRs contain a prominent central splay (Fig. 4) which is solvent filled and accessible from the extracellular side. This central splay is notably absent from class A GPCRs (Fig. 5) , represents a tantalizing target for agonists/antagonists, and is the focus of much current research into GCGR signal regulation. [3] 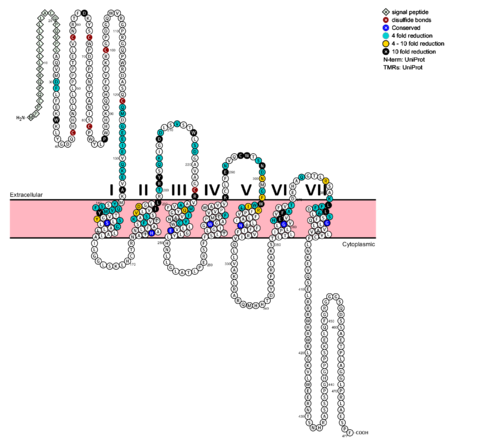 Fig. 6: Snake Plot of GCGR TMD[5] The snake plot (Fig. 6) shows the conservation and effects of mutagenesis in the 7TMD structure of class B human GPCR. The highly conserved amino acids imply an importance to that functioning of the individual residues and their interactions. The amino acids which have a great impact on the function of the receptor are highlighted in teal, yellow, and black, and offer evidence that the position and interaction of the amino acid is crucial for protein function. Most of the residues that play an important role in glucagon binding face the main cavity of the 7TM structure. Mutagenesis in these positions highly compromises the functioning of the glucagon binding. Functions of Glucagon receptor (GCGR)It has been discovered that the large, soluble N-terminal extracellular domains (ECD) of GCGR are primary in ligand selectivity with the deep, ligand pocket (Fig. 7) of the TMD providing secondary recognition. [6] Because of the difficulty of stabilizing and crystallizing Class B TMDs, very little is known about the conformational changes that transduce cell signals endogenously. It is known that GCGR can regulate additional signal pathways including G-proteins of the Gαi family through the adoption of differing receptor conformations. Research is ongoing. [8] GCGR generates downstream signals predominantly through the increase of intracellular cAMP, however there are other pathways being uncovered that are the result of GCGR adopting multiple, active conformations. Researchers are currently investigating how receptor activity-modifying proteins (RAMPs) interact with the ligand and GCGR in which the signaling bias of the receptor is altered. [9] GPCR activity is regularly quantified by ligand binding affinity, potency, efficacy, and kinetics. These measurement are used to measure drug ligand interactions in vivo. Recently, GPCRs have been crystallized and catalogued, which tend to include a need to stabilize the receptor, emphasizing the instability of the G coupled protein receptor. Zhang et. al. imply the importance of receptor folding in the cell membrane, in the human class B GPCR the 7TM portion, for receptor stability and function. [10] The Signal Peptide: GlucagonGlucagon, a signaling ligand in the metabolic pathway, has three main biological functions. Glucagon is a regulator of the production of cholesterol, which is an energetically intensive process. When energy resources are low, downregulation of cholesterol production begins with glucagon binding to GCGR, which stimulates the phosphorylation of HMG-CoA. Once HMG-CoA has been phosphorylated, it is inactivated and cholesterol production is moderated to conserve energy. Glucagon also takes part in fatty acid mobilization by affecting levels of adipose tissue in the organism. Activation of GCGR by glucagon initiates triacylglycerol breakdown and the phosphorylation of perilipin and lipases via cAMP signal pathways. This allows the body to export fatty acids to the liver and other crucial tissues for energy use and makes more glucose available for use in brain functioning. Glucagon's main role is the regulation of blood glucose levels. Glucagon lowers the concentration of fructose 2,6-bisphosphate which is an allosteric inhibitor of the gluconeogenic enzyme fructose 1,6-bisphosphotase and activates phosphofructose kinase 1, which increases glucose levels via glycolysis. Essential, conserved residues of glucagon, as discovered through mutagenesis and photo cross-linking studies have been labeled and colored in red. [5] Through mutagenesis and photo-crosslinking studies, several residues deep within the central cavity of the GCGR 7TMD were discovered neighboring Glu362, which is approximately 19 angstroms from the base of the EC stalk and the location of Tyr138. (Fig. 8)
Four essential residues exist deep within the central cavity which all play strong roles in ligand binding affinity. (Fig. 9)
A narrow entry gives way to a large, anchoring site for residues 1-4 of glucagon. (Fig. 10) Essential to glucagon's binding, a long, N-terminal tail winds to a clump of 4 residues, culminating in bulge that fits into the central, anchoring site of the 7TMD. (Fig. 11) Clinical relevanceClass B secretin-like receptors have gained relevance in therapeutics and drug targets. Maintaining information about the class B GPCRs conformational flexibility, allows for a better understanding of the receptor-ligand binding and its pharmaceutical relevance. The 7TM structure offers a direct connect between the extracellular and intracellular region, which offers a mechanism for signal transduction within the cell. GPCRs regulate cellular processes as required by the organs in which they are located. GPCR’s are used in the functioning of neuron synapses, ion transport regulation, homeostasis, cell division, and cell morphology. Mutations in the GPCR have been linked with retinitis pigmentosa, female infertility, nephrogenic diabetes insipidus, and familial exudative vitreoretinopathy. [12] A variety of small molecule modulators have been developed over the past several years providing the promise of enhanced pharmaceutical regulation of GCGR. [6](Fig's. 12 and 13)  Fig. 12: Small molecule regulators of GCGR, part 1[6].  Fig. 13: Small molecule regulators of GCGR, part 2[6]. Utilizing the visualizations of the GCGR 7TMD and glucagon peptide ligand, dimensional/structural analyses can be performed to develop models for novel molecules of increasing specificity for GCGR binding/regulation. Performing a dimensional analysis between the binding pocket and the base of the EC stalk, a large pseudopeptide molecule of 17-24 angstroms in size could be utilized to mimic the characteristics of GCGR's natural ligand, glucagon. (Fig's. 14 and 15) See Also
References
| ||||||||||||