This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Brittany Stankavich/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 17: | Line 17: | ||
== Structure == | == Structure == | ||
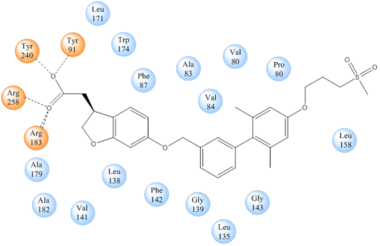
| - | hGPR40 is composed of seven-transmembrane helices that are characteristic of G-protein coupled receptors (GPCR). While there is relatively low sequence identity between hGPR40 and peptide-binding | + | hGPR40 is composed of seven-transmembrane helices that are characteristic of G-protein coupled receptors (GPCR)<ref name="crystal">PMID: 25043059</ref>. While there is relatively low sequence identity between hGPR40 and peptide-binding and opioid GPCRs, they do share structural similarities such as a conserved <scene name='72/727085/Hairpin_loop/3'>hairpin loop</scene> motif on <scene name='72/727085/Ecl2/3'>extracellular loop 2 </scene>(ECL2)<ref name="crystal"/>. In addition, there is a conserved <scene name='72/727085/Disulfide/2'>disulphide bond</scene> that is formed between transmembrane helix 3 (Cys 79) and the C-terminus of ECL2 (Cys170)<ref name="crystal"/>. Compared to peptide-binding and opioid GPCRs which have distinctive β-sheets spanning from transmembrane helix 4 to 5, hGPR40 possesses a shorter B-sheet-like region which has low B-factors<ref name="crystal"/>. This reflects the low mobility of the region that limits the overall flexibility of the adjacent portion of ECL2 between Leu171 and Asp175<ref name="crystal"/>. A unique feature of hGPR40 is the presence of an additional 13 residues (Pro147 to Gly159) on ECL2 which is absent on all the other peptide/opioid receptors<ref name="crystal"/>. These extra residues form a separate <scene name='72/727085/Auxiliary_loop/2'>auxiliary loop</scene> between the B-sheet-like region and transmembrane 4. Together, the auxiliary loop and ECL2 of hGPR40 function as a <scene name='72/727085/Ecl2_cap/2'>roof </scene> over the canonical binding site covering it from the central extracellular region<ref name="crystal"/>. |
| + | The canonical binding pocket for many other GPCRs is solvent exposed and centrally located between the transmembrane helices allowing ligands to directly bind from the extracellular space<ref name="crystal"/>. However, because the ECL2 acts as a roof to this canonical binding site, it inhibits ligands from entering directly from the extracellular region. Instead, the highly lipophilic nature of hGPRC40’s ligands allow it to enter a <scene name='72/727085/Hgpr40_entry/2'>noncanonical binding pocket </scene> between TM3 and TM4 by moving through the lipid bilayer<ref name="crystal"/>. | ||
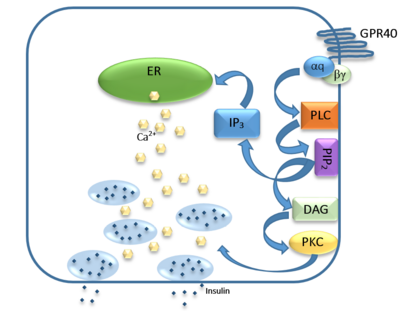
== Ligand Binding == | == Ligand Binding == | ||
Revision as of 12:39, 21 April 2016
- User:Brittany Stankavich/Sandbox 1
hGPR40 Homo sapiens
| |||||||||||
References
- ↑ Ren XM, Cao LY, Zhang J, Qin WP, Yang Y, Wan B, Guo LH. Investigation of the Binding Interaction of Fatty Acids with Human G Protein-Coupled Receptor 40 Using a Site-Specific Fluorescence Probe by Flow Cytometry. Biochemistry. 2016 Mar 17. PMID:26974599 doi:http://dx.doi.org/10.1021/acs.biochem.6b00079
- ↑ 2.0 2.1 2.2 2.3 2.4 Ichimura A, Hirasawa A, Hara T, Tsujimoto G. Free fatty acid receptors act as nutrient sensors to regulate energy homeostasis. Prostaglandins Other Lipid Mediat. 2009 Sep;89(3-4):82-8. doi:, 10.1016/j.prostaglandins.2009.05.003. Epub 2009 May 19. PMID:19460454 doi:http://dx.doi.org/10.1016/j.prostaglandins.2009.05.003
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 Srivastava A, Yano J, Hirozane Y, Kefala G, Gruswitz F, Snell G, Lane W, Ivetac A, Aertgeerts K, Nguyen J, Jennings A, Okada K. High-resolution structure of the human GPR40 receptor bound to allosteric agonist TAK-875. Nature. 2014 Jul 20. doi: 10.1038/nature13494. PMID:25043059 doi:http://dx.doi.org/10.1038/nature13494
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 Morgan NG, Dhayal S. G-protein coupled receptors mediating long chain fatty acid signalling in the pancreatic beta-cell. Biochem Pharmacol. 2009 Dec 15;78(12):1419-27. doi: 10.1016/j.bcp.2009.07.020., Epub 2009 Aug 4. PMID:19660440 doi:http://dx.doi.org/10.1016/j.bcp.2009.07.020
- ↑ Ren XM, Cao LY, Zhang J, Qin WP, Yang Y, Wan B, Guo LH. Investigation of the Binding Interaction of Fatty Acids with Human G Protein-Coupled Receptor 40 Using a Site-Specific Fluorescence Probe by Flow Cytometry. Biochemistry. 2016 Mar 17. PMID:26974599 doi:http://dx.doi.org/10.1021/acs.biochem.6b00079
- ↑ Hanson MA, Roth CB, Jo E, Griffith MT, Scott FL, Reinhart G, Desale H, Clemons B, Cahalan SM, Schuerer SC, Sanna MG, Han GW, Kuhn P, Rosen H, Stevens RC. Crystal structure of a lipid G protein-coupled receptor. Science. 2012 Feb 17;335(6070):851-5. PMID:22344443 doi:10.1126/science.1215904
- ↑ Li X, Zhong K, Guo Z, Zhong D, Chen X. Fasiglifam (TAK-875) Inhibits Hepatobiliary Transporters: A Possible Factor Contributing to Fasiglifam-Induced Liver Injury. Drug Metab Dispos. 2015 Nov;43(11):1751-9. doi: 10.1124/dmd.115.064121. Epub 2015, Aug 14. PMID:26276582 doi:http://dx.doi.org/10.1124/dmd.115.064121
- ↑ 8.0 8.1 Takano R, Yoshida M, Inoue M, Honda T, Nakashima R, Matsumoto K, Yano T, Ogata T, Watanabe N, Hirouchi M, Yoneyama T, Ito S, Toda N. Discovery of DS-1558: A Potent and Orally Bioavailable GPR40 Agonist. ACS Med Chem Lett. 2015 Jan 13;6(3):266-70. doi: 10.1021/ml500391n. eCollection, 2015 Mar 12. PMID:25815144 doi:http://dx.doi.org/10.1021/ml500391n