This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1hvx
From Proteopedia
| Line 4: | Line 4: | ||
|PDB= 1hvx |SIZE=350|CAPTION= <scene name='initialview01'>1hvx</scene>, resolution 2.0Å | |PDB= 1hvx |SIZE=350|CAPTION= <scene name='initialview01'>1hvx</scene>, resolution 2.0Å | ||
|SITE= | |SITE= | ||
| - | |LIGAND= <scene name='pdbligand=CA:CALCIUM+ION'>CA</scene> | + | |LIGAND= <scene name='pdbligand=CA:CALCIUM+ION'>CA</scene>, <scene name='pdbligand=NA:SODIUM+ION'>NA</scene> |
| - | |ACTIVITY= [http://en.wikipedia.org/wiki/Alpha-amylase Alpha-amylase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=3.2.1.1 3.2.1.1] | + | |ACTIVITY= <span class='plainlinks'>[http://en.wikipedia.org/wiki/Alpha-amylase Alpha-amylase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=3.2.1.1 3.2.1.1] </span> |
|GENE= AMYT631 ([http://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Info&srchmode=5&id=1422 Geobacillus stearothermophilus]) | |GENE= AMYT631 ([http://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Info&srchmode=5&id=1422 Geobacillus stearothermophilus]) | ||
| + | |DOMAIN= | ||
| + | |RELATEDENTRY= | ||
| + | |RESOURCES=<span class='plainlinks'>[http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=1hvx FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1hvx OCA], [http://www.ebi.ac.uk/pdbsum/1hvx PDBsum], [http://www.rcsb.org/pdb/explore.do?structureId=1hvx RCSB]</span> | ||
}} | }} | ||
| Line 28: | Line 31: | ||
[[Category: Suvd, D.]] | [[Category: Suvd, D.]] | ||
[[Category: Takase, K.]] | [[Category: Takase, K.]] | ||
| - | [[Category: | + | [[Category: alpha-1,4-glucan-4-glucanohydrolase]] |
| - | + | [[Category: alpha-amylase,starch degradation]] | |
| - | + | ||
| - | + | ||
| - | [[Category: alpha-amylase]] | + | |
[[Category: calcium]] | [[Category: calcium]] | ||
[[Category: glycosyltransferase]] | [[Category: glycosyltransferase]] | ||
[[Category: hydrolase]] | [[Category: hydrolase]] | ||
[[Category: sodium]] | [[Category: sodium]] | ||
| - | [[Category: starch degradation]] | ||
[[Category: thermostability]] | [[Category: thermostability]] | ||
| - | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Sun Mar 30 21:11:09 2008'' |
Revision as of 18:11, 30 March 2008
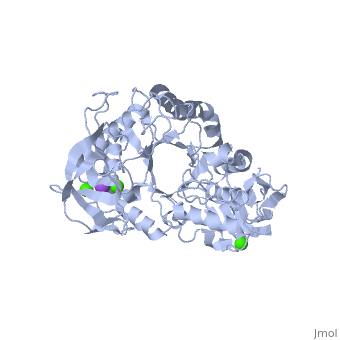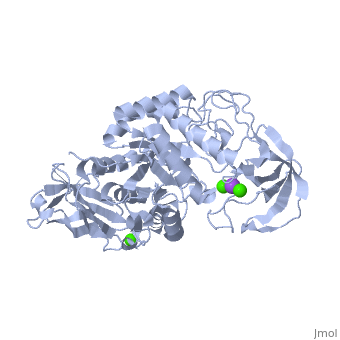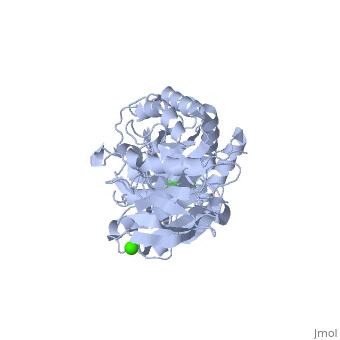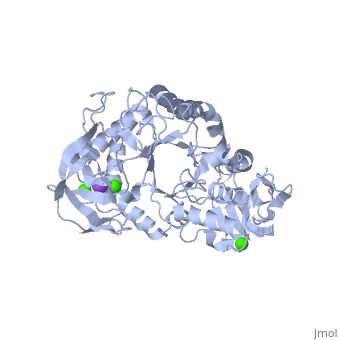
| |||||||
| , resolution 2.0Å | |||||||
|---|---|---|---|---|---|---|---|
| Ligands: | , | ||||||
| Gene: | AMYT631 (Geobacillus stearothermophilus) | ||||||
| Activity: | Alpha-amylase, with EC number 3.2.1.1 | ||||||
| Resources: | FirstGlance, OCA, PDBsum, RCSB | ||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||
BACILLUS STEAROTHERMOPHILUS ALPHA-AMYLASE
Overview
The crystal structure of a thermostable alpha-amylase from Bacillus stearothermophilus (BSTA) has been determined at 2.0 A resolution. The main-chain fold is almost identical to that of the known crystal structure of Bacillus licheniformis alpha-amylase (BLA). BLA is known to be more stable than BSTA. A structural comparison between the crystal structures of BSTA and BLA showed significant differences that may account for the difference in their thermostabilities, as follows. (i) The two-residue insertion in BSTA, Ile181-Gly182, pushes away the spatially contacting region including Asp207, which corresponds to Ca(2+)-coordinating Asp204 in BLA. As a result, Asp207 cannot coordinate the Ca(2+). (ii) BSTA contains nine fewer hydrogen bonds than BLA, which costs about 12 kcal/mol. This tendency is prominent in the (beta/alpha)(8)-barrel, where 10 fewer hydrogen bonds were observed in BSTA. BLA forms a denser hydrogen bond network in the inter-helical region, which may stabilize alpha-helices in the barrel. (iii) A few small voids observed in the alpha-helical region of the (beta/alpha)(8)-barrel in BSTA decrease inter-helical compactness and hydrophobic interactions. (iv) The solvent-accessible surface area of charged residues in BLA is about two times larger than that in BSTA.
About this Structure
1HVX is a Single protein structure of sequence from Geobacillus stearothermophilus. Full crystallographic information is available from OCA.
Reference
Crystal structure of Bacillus stearothermophilus alpha-amylase: possible factors determining the thermostability., Suvd D, Fujimoto Z, Takase K, Matsumura M, Mizuno H, J Biochem. 2001 Mar;129(3):461-8. PMID:11226887
Page seeded by OCA on Sun Mar 30 21:11:09 2008