This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Telomerase
From Proteopedia
(Difference between revisions)
| Line 27: | Line 27: | ||
The reverse transcription process begins by the telomerase RNA binding to the 3' end of the overhanging DNA sequence. Once primed, the G-rich sequence (~6-8 nucleotides) polymerizes the end of the DNA. The RNA primer then translocates down the new DNA sequence and polymerizes again. This process repeats until the entire telomere is formed. After one side of the telomere is formed, the telomerase attaches the complementary nucleotide bases to complete the double-stranded telomere at the end of the DNA double helix. | The reverse transcription process begins by the telomerase RNA binding to the 3' end of the overhanging DNA sequence. Once primed, the G-rich sequence (~6-8 nucleotides) polymerizes the end of the DNA. The RNA primer then translocates down the new DNA sequence and polymerizes again. This process repeats until the entire telomere is formed. After one side of the telomere is formed, the telomerase attaches the complementary nucleotide bases to complete the double-stranded telomere at the end of the DNA double helix. | ||
| - | + | </StructureSection> | |
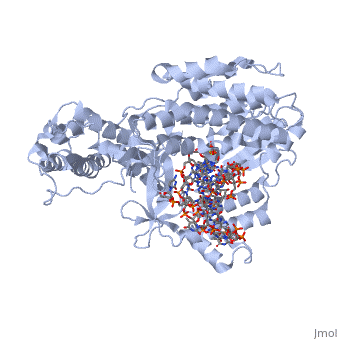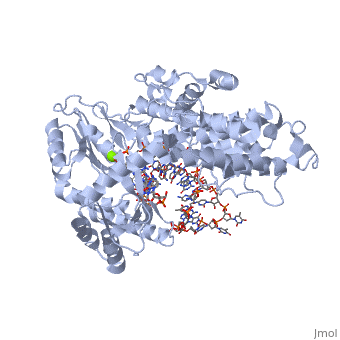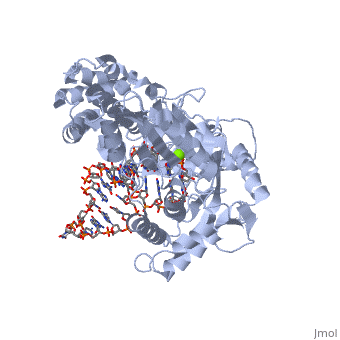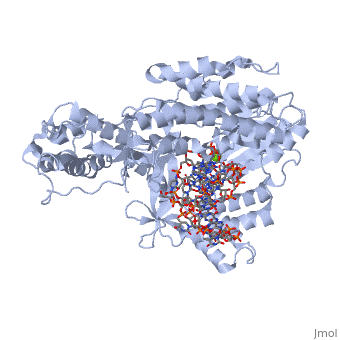
==Telomerase 3D structures== | ==Telomerase 3D structures== | ||
Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | ||
Revision as of 08:14, 8 September 2016
| |||||||||||
Telomerase 3D structures
Updated on 08-September-2016
References
- ↑ 1.0 1.1 Blackburn EH. Telomerases. Annu Rev Biochem. 1992;61:113-29. PMID:1497307 doi:http://dx.doi.org/10.1146/annurev.bi.61.070192.000553
- ↑ 2.0 2.1 Blackburn EH. Telomeres and telomerase: the means to the end (Nobel lecture). Angew Chem Int Ed Engl. 2010 Oct 4;49(41):7405-21. doi: 10.1002/anie.201002387. PMID:20821774 doi:http://dx.doi.org/10.1002/anie.201002387
- ↑ 3.0 3.1 3.2 3.3 3.4 Corey DR. Telomeres and telomerase: from discovery to clinical trials. Chem Biol. 2009 Dec 24;16(12):1219-23. doi: 10.1016/j.chembiol.2009.12.001. PMID:20064431 doi:http://dx.doi.org/10.1016/j.chembiol.2009.12.001
- ↑ 4.0 4.1 4.2 Gillis AJ, Schuller AP, Skordalakes E. Structure of the Tribolium castaneum telomerase catalytic subunit TERT. Nature. 2008 Oct 2;455(7213):633-7. Epub 2008 Aug 31. PMID:18758444 doi:http://dx.doi.org/10.1038/nature07283
- ↑ 5.0 5.1 Mitchell M, Gillis A, Futahashi M, Fujiwara H, Skordalakes E. Structural basis for telomerase catalytic subunit TERT binding to RNA template and telomeric DNA. Nat Struct Mol Biol. 2010 Apr;17(4):513-8. Epub 2010 Mar 28. PMID:20357774 doi:10.1038/nsmb.1777
</StructureSection>