This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
TGF-beta receptor
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
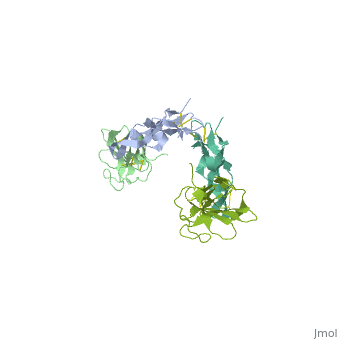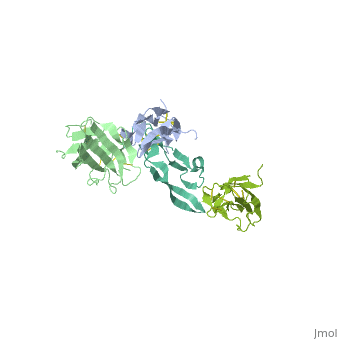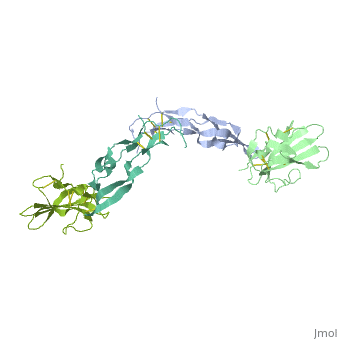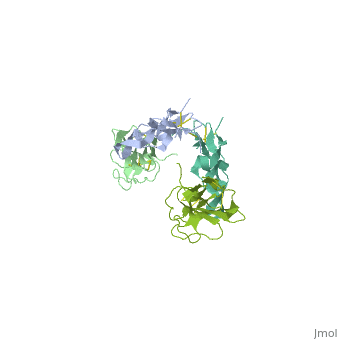
<StructureSection load='1ktz' size='340' side='right' caption='Human hTGFBR-II extracellular domain (green) complex with TGF-β3 (grey) (PDB code [[1ktz]])' scene=''> | <StructureSection load='1ktz' size='340' side='right' caption='Human hTGFBR-II extracellular domain (green) complex with TGF-β3 (grey) (PDB code [[1ktz]])' scene=''> | ||
| + | __TOC__ | ||
== Function == | == Function == | ||
| - | '''TGF-β receptors''' (Transforming Growth Factor) (TGFBR) are serine/threonine kinase receptors. They are involved in paracrine signaling and are found in many types of tissue. TGF-β ligands include bone morphogenetic proteins, growth and initiation factors, anti-Mullerian hormone, activin, nodal TGF-β. | + | '''TGF-β receptors''' (Transforming Growth Factor) (TGFBR) are serine/threonine kinase receptors. They are involved in paracrine signaling and are found in many types of tissue. TGF-β ligands include bone morphogenetic proteins, growth and initiation factors, anti-Mullerian hormone, activin, nodal TGF-β<ref>PMID:21294712</ref>. There are 3 types of TGFBR: <br /> |
*'''TGFBR I''' forms heteromeric complex with TGFBR II when it is bound to TGF-β. The complex transduces the TGF-β signal from the cell surface to the cytoplasm by phosphorylating proteins which regulate the transcription of genes related to cell proliferation. TGFBR I has high affinity for TGF-β1 and low affinity for TGF-β2. <br /> | *'''TGFBR I''' forms heteromeric complex with TGFBR II when it is bound to TGF-β. The complex transduces the TGF-β signal from the cell surface to the cytoplasm by phosphorylating proteins which regulate the transcription of genes related to cell proliferation. TGFBR I has high affinity for TGF-β1 and low affinity for TGF-β2. <br /> | ||
*'''TGFBR II''' is a tumor suppressor transmembrane protein. TGFBR II has high affinity for TGF-β1 and low affinity for TGF-β2. <br /> | *'''TGFBR II''' is a tumor suppressor transmembrane protein. TGFBR II has high affinity for TGF-β1 and low affinity for TGF-β2. <br /> | ||
| Line 8: | Line 9: | ||
== Disease == | == Disease == | ||
| - | Over-expression of TGF causes kidney disease, diabetes and renal disease. Mutations in TGFBR II cause various types of tumors. | + | Over-expression of TGF causes kidney disease, diabetes and renal disease. Mutations in TGFBR II cause various types of tumors<ref>PMID:23884466</ref>. |
== Structural highlights == | == Structural highlights == | ||
| + | TGFBR structure contains a 100-140 residues ligand-binding N-terminal extracellular domain; a transmembrane domain; a 350-400 amino acid cytoplasmic kinase domain; and a C-terminal zona pellucida (ZP) domain of ca 260 residues which has a role in protein polymerization. | ||
</StructureSection> | </StructureSection> | ||
Revision as of 07:48, 11 September 2016
| |||||||||||
3D Structures of TGF-β receptor
Updated on 11-September-2016
References
- ↑ Koudelakova T, Chovancova E, Brezovsky J, Monincova M, Fortova A, Jarkovsky J, Damborsky J. Substrate specificity of haloalkane dehalogenases. Biochem J. 2011 Apr 15;435(2):345-54. doi: 10.1042/BJ20101405. PMID:21294712 doi:http://dx.doi.org/10.1042/BJ20101405
- ↑ Frischmeyer-Guerrerio PA, Guerrerio AL, Oswald G, Chichester K, Myers L, Halushka MK, Oliva-Hemker M, Wood RA, Dietz HC. TGFbeta receptor mutations impose a strong predisposition for human allergic disease. Sci Transl Med. 2013 Jul 24;5(195):195ra94. doi: 10.1126/scitranslmed.3006448. PMID:23884466 doi:http://dx.doi.org/10.1126/scitranslmed.3006448