This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Transfer RNA (tRNA)
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
<StructureSection load='' size='350' side='right' scene='1ehz/1ehz_default/3' caption='Yeast Phe-tRNA (PDB code [[1ehz]])'> | <StructureSection load='' size='350' side='right' scene='1ehz/1ehz_default/3' caption='Yeast Phe-tRNA (PDB code [[1ehz]])'> | ||
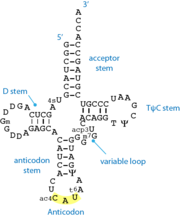
[[Image:TRNA.png|left|thumb|Standard 2D cloverleaf structure of tRNA. The shown example is methionine-specific tRNA from ''E.coli'' ]] | [[Image:TRNA.png|left|thumb|Standard 2D cloverleaf structure of tRNA. The shown example is methionine-specific tRNA from ''E.coli'' ]] | ||
| + | __TOC__ | ||
| + | ==Function== | ||
'''tRNA''' or '''transfer RNA''' is stable, structured RNA present in all living cells. tRNA participates in the process of protein [[translation]] by the [[ribosome]]. Varying tRNA molecules carry a specific amino acid esterified on their 3'-OH group (the acceptor end). They also carry a specific triplet sequence, the '''anticodon''', which pairs with its complementary '''codon''' on the messenger RNA, within the ribosome. | '''tRNA''' or '''transfer RNA''' is stable, structured RNA present in all living cells. tRNA participates in the process of protein [[translation]] by the [[ribosome]]. Varying tRNA molecules carry a specific amino acid esterified on their 3'-OH group (the acceptor end). They also carry a specific triplet sequence, the '''anticodon''', which pairs with its complementary '''codon''' on the messenger RNA, within the ribosome. | ||
| Line 11: | Line 13: | ||
==Modified nucleotides== | ==Modified nucleotides== | ||
| - | Most tRNAs contain modified nucleotides<ref>PMID:20459084</ref>, which are added post-transcriptionally by specific enzymes. Common modifications include isomerisation of uridines into pseudouridines (Ψ), methylation of either the ribose and/or the base, thiolation, reduction of uridines into dihydrouridines (D). The anticodon loop of the tRNA quite often contains hypermodified bases, the function of which is to | + | Most tRNAs contain modified nucleotides<ref>PMID:20459084</ref>, which are added post-transcriptionally by specific enzymes. Common modifications include isomerisation of uridines into pseudouridines (Ψ), methylation of either the ribose and/or the base, thiolation, reduction of uridines into dihydrouridines (D). The anticodon loop of the tRNA quite often contains hypermodified bases, the function of which is to stabilize the codon-anticodon interaction within the ribosome. The nature and position of nucleotide modifications is both specific of the organism and the tRNA type. |
Common modified nucleotides include : | Common modified nucleotides include : | ||
* <scene name='TRNA/Fullview_methylu/2'>5-methyluridine </scene>(ribothymidine) at position 54 | * <scene name='TRNA/Fullview_methylu/2'>5-methyluridine </scene>(ribothymidine) at position 54 | ||
Revision as of 06:59, 25 September 2016
| |||||||||||
3D Structures of tRNA
Updated on 25-September-2016
See Also
References
- ↑ Monajemi H, Omar NY, Daud MN, Zain SM, Abdullah WA. The role of initiator tRNAimet in fidelity of initiation of protein synthesis. Nucleosides Nucleotides Nucleic Acids. 2011 Sep;30(9):726-39. doi:, 10.1080/15257770.2011.605780. PMID:21902474 doi:http://dx.doi.org/10.1080/15257770.2011.605780
- ↑ Motorin Y, Helm M. tRNA stabilization by modified nucleotides. Biochemistry. 2010 Jun 22;49(24):4934-44. PMID:20459084 doi:10.1021/bi100408z
Reference for the structure
- Shi H, Moore PB. The crystal structure of yeast phenylalanine tRNA at 1.93 A resolution: a classic structure revisited. RNA. 2000 Aug;6(8):1091-105. PMID:10943889
Proteopedia Page Contributors and Editors (what is this?)
Karsten Theis, Wayne Decatur, Michal Harel, Frédéric Dardel, Ann Taylor, Joel L. Sussman, Alexander Berchansky
Categories: Trna | Topic Page | Translation | Modification | RNA | Amino acid