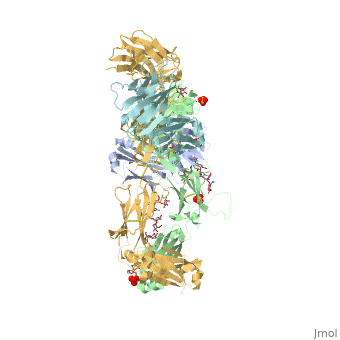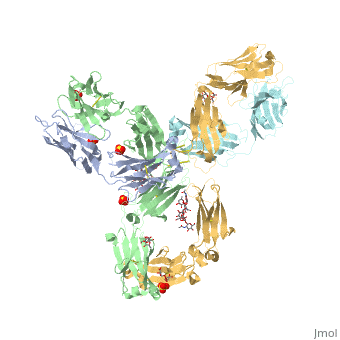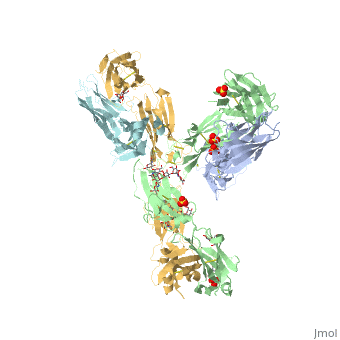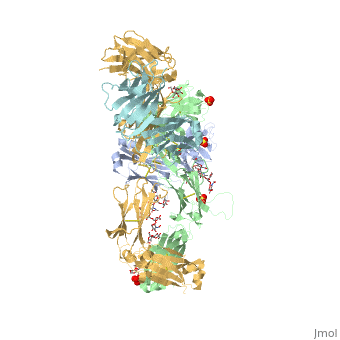This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Keytruda
From Proteopedia
(Difference between revisions)
| Line 12: | Line 12: | ||
===PD-L1/PD-1 Interaction=== | ===PD-L1/PD-1 Interaction=== | ||
The complex formed when protein-derived ligand, PD-L1, interacts with the inhibitory receptor, PD-1, suppresses immune responses against autoantigens and helps in peripheral immune tolerance. However, when tumors over express PD-L1, the interaction with PD-1 inhibits T-lymphocyte proliferation, release of cytokines, and cytotoxicity, exhausting tumor-specific T-cells. There are a total of 12 PD-1<sub>ECD</sub> residues that are involved in forming the complex with the N-terminal half of PD-1<sub>ECD</sub> (PD-1<sub>ECD-N</sub>). Nine hydrogen bonds, 3 water-mediated hydrogen bonds, 2 salt bridges, and numerous hydrophobic interactions make up the PD-1<sub>ECD</sub>/PD-1<sub>ECD-N</sub> interaction. The CC’FG sheet within both proteins is the main interaction point. A hydrophobic surface patch is formed when the PD-1<sub>ECD</sub> is in complex with PD-L1<sub>ECD-N</sub>. The PD-1<sub>ECD</sub> residues involved include Val64, Tyr68, Ile126, Leu128, Ala132 and Ile134. Numerous [http://www.nature.com/articles/srep35297/figures/1 Hydrophilic amino acids] that encircle PD-L1<sub>ECD-N</sub> form salt bridges and hydrogen bonds with Asn66, Tyr68, Gln75, Thr76, Asp77, Lys78, Ala132 and Glu136 of PD-1<sub>ECD</sub> <ref name="horita" />. | The complex formed when protein-derived ligand, PD-L1, interacts with the inhibitory receptor, PD-1, suppresses immune responses against autoantigens and helps in peripheral immune tolerance. However, when tumors over express PD-L1, the interaction with PD-1 inhibits T-lymphocyte proliferation, release of cytokines, and cytotoxicity, exhausting tumor-specific T-cells. There are a total of 12 PD-1<sub>ECD</sub> residues that are involved in forming the complex with the N-terminal half of PD-1<sub>ECD</sub> (PD-1<sub>ECD-N</sub>). Nine hydrogen bonds, 3 water-mediated hydrogen bonds, 2 salt bridges, and numerous hydrophobic interactions make up the PD-1<sub>ECD</sub>/PD-1<sub>ECD-N</sub> interaction. The CC’FG sheet within both proteins is the main interaction point. A hydrophobic surface patch is formed when the PD-1<sub>ECD</sub> is in complex with PD-L1<sub>ECD-N</sub>. The PD-1<sub>ECD</sub> residues involved include Val64, Tyr68, Ile126, Leu128, Ala132 and Ile134. Numerous [http://www.nature.com/articles/srep35297/figures/1 Hydrophilic amino acids] that encircle PD-L1<sub>ECD-N</sub> form salt bridges and hydrogen bonds with Asn66, Tyr68, Gln75, Thr76, Asp77, Lys78, Ala132 and Glu136 of PD-1<sub>ECD</sub> <ref name="horita" />. | ||
| - | == Disease in Humans | + | == Disease in Humans == |
T-cells are a major component of the immune response in the human body. They have the ability to recognize cancer-related antigens as non-self and eliminate those cells <ref>doi 10.2147/DDDT.S78036</ref>. PD-L1 and PD-L2 are ligands expressed by some tumors and inhibit T-cell function when bound to PD-1, which is located on the surface of antigen-specific T-cells <ref>doi 10.1007/s40265-016-0543-x</ref>. When PD-L1 is ligated to PD-1 an adaptive immune response occurs, and this allows cancer cells to bypass immune surveillance and grow uncontrollably. Pembrolizumab is an FDA-approved treatment that works as a PD-1 pathway inhibitor to fight numerous forms of cancer, such as metastatic melanoma and non-small cell lung cancer. As an inhibitor, Pembrolizumab targets the cell death of PD-1 and blocks the immune checkpoint pathway. Pembrolizumab has a very high affinity to PD-1, allowing it to block the interaction between PD-1 with PD-L1 and PD-L2 very efficiently. It antagonizes the interaction between PD-1 and its known ligands, and re-activates anti-tumor immunity <ref name="log" />. The PD-1/PD-L1 interaction inhibits T-lymphocyte proliferation, releases cytokines and cytotoxicity, and exhausts tumor-specific T-cells. The inhibition of this pathway reverses the exhausted t-cell phenotype and normalizes the anti-tumor response. One downside of Pembrolizumab is that it may cause inflammatory side effects <ref name="horita" />. | T-cells are a major component of the immune response in the human body. They have the ability to recognize cancer-related antigens as non-self and eliminate those cells <ref>doi 10.2147/DDDT.S78036</ref>. PD-L1 and PD-L2 are ligands expressed by some tumors and inhibit T-cell function when bound to PD-1, which is located on the surface of antigen-specific T-cells <ref>doi 10.1007/s40265-016-0543-x</ref>. When PD-L1 is ligated to PD-1 an adaptive immune response occurs, and this allows cancer cells to bypass immune surveillance and grow uncontrollably. Pembrolizumab is an FDA-approved treatment that works as a PD-1 pathway inhibitor to fight numerous forms of cancer, such as metastatic melanoma and non-small cell lung cancer. As an inhibitor, Pembrolizumab targets the cell death of PD-1 and blocks the immune checkpoint pathway. Pembrolizumab has a very high affinity to PD-1, allowing it to block the interaction between PD-1 with PD-L1 and PD-L2 very efficiently. It antagonizes the interaction between PD-1 and its known ligands, and re-activates anti-tumor immunity <ref name="log" />. The PD-1/PD-L1 interaction inhibits T-lymphocyte proliferation, releases cytokines and cytotoxicity, and exhausts tumor-specific T-cells. The inhibition of this pathway reverses the exhausted t-cell phenotype and normalizes the anti-tumor response. One downside of Pembrolizumab is that it may cause inflammatory side effects <ref name="horita" />. | ||
Revision as of 23:45, 5 December 2016
Pembrolizumab antibody against programmed cell death-1 receptor
| |||||||||||
References
- ↑ 1.0 1.1 Longoria TC, Tewari KS. Evaluation of the pharmacokinetics and metabolism of pembrolizumab in the treatment of melanoma. Expert Opin Drug Metab Toxicol. 2016 Oct;12(10):1247-53. doi:, 10.1080/17425255.2016.1216976. Epub 2016 Aug 16. PMID:27485741 doi:http://dx.doi.org/10.1080/17425255.2016.1216976
- ↑ 2.0 2.1 2.2 2.3 Horita S, Nomura Y, Sato Y, Shimamura T, Iwata S, Nomura N. High-resolution crystal structure of the therapeutic antibody pembrolizumab bound to the human PD-1. Sci Rep. 2016 Oct 13;6:35297. doi: 10.1038/srep35297. PMID:27734966 doi:http://dx.doi.org/10.1038/srep35297
- ↑ Rajakulendran T, Adam DN. Spotlight on pembrolizumab in the treatment of advanced melanoma. Drug Des Devel Ther. 2015 Jun 4;9:2883-6. doi: 10.2147/DDDT.S78036. eCollection, 2015. PMID:26082618 doi:http://dx.doi.org/10.2147/DDDT.S78036
- ↑ Deeks ED. Pembrolizumab: A Review in Advanced Melanoma. Drugs. 2016 Mar;76(3):375-86. doi: 10.1007/s40265-016-0543-x. PMID:26846323 doi:http://dx.doi.org/10.1007/s40265-016-0543-x