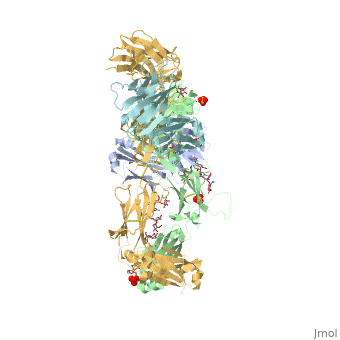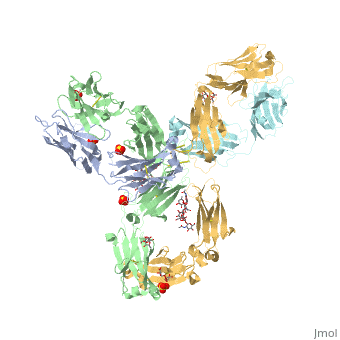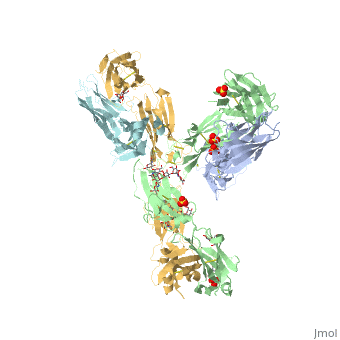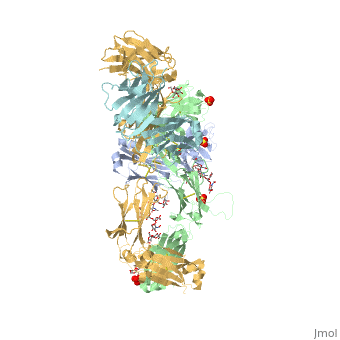This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Keytruda
From Proteopedia
(Difference between revisions)
| Line 6: | Line 6: | ||
== Mechanism == | == Mechanism == | ||
| - | + | ||
===Pembrolizumab/PD-1 Interaction=== | ===Pembrolizumab/PD-1 Interaction=== | ||
| - | In order for Pembrolizumab to block PD-1, Pembrolizumab forms a large, flat paratope (antigen-binding site) that can sustain PD-1’s large epitope (where antibody attaches on antigen). The induced interaction between Pembrolizumab and PD-1 gives rise to a surface conformational change on PD-1. The new structure of PD-1 becomes a very shallow, “crescent”-like shape, in contrast to the flat conformation when bound to PD-L1 <ref name="horita">DOI:10.1038/srep35297</ref>. | + | In order for Pembrolizumab to block PD-1, Pembrolizumab forms a large, flat paratope (antigen-binding site) that can sustain PD-1’s large epitope (where antibody attaches on antigen). The induced [http://www.nature.com/articles/srep35297/figures/2 interaction between Pembrolizumab and PD-1] gives rise to a surface conformational change on PD-1. The new structure of PD-1 becomes a very shallow, “crescent”-like shape, in contrast to the flat conformation when bound to PD-L1 <ref name="horita">DOI:10.1038/srep35297</ref>. |
===PemFv/PD-1 Interaction=== | ===PemFv/PD-1 Interaction=== | ||
The Fv fragment of Pembrolizumab can form a complex with the extracellular domain (ECD) of PD-1. Both PemFv and PD-1<sub>ECD</sub> contain interchain disulfide bonds. PemFv interacts predominantly in the major groove of PD-1, which is formed on one surface by the CC’FG antiparallel β−sheet and the BC, C’D, and FG loops. There are 15 direct hydrogen bonds between the residues, 15 water-mediated hydrogen bonds, 2 salt bridges, and many hydrophobic interactions. There are a total of 26 PD-1<sub>ECD</sub> residues involved in the interaction with PemFv, with residues in loop C’D (Pro84 to Gly90) and strand C’ (Gln75 to Lys 78) playing a major role. These key components of PD-1 mainly form interactions through salt bridges and hydrogen bonds with complementary determining regions, the variable domains, of Pembrolizumab. <scene name='74/745945/Chain_b_amino_acids/1'>Thr30, Tyr33, Ser54, Tyr101, Arg102</scene> on Chain B of Pembrolizumab form bonds with Asp77, Gln75, Lys78, Thr76, Tyr68, and Asn66 of PD-1. It is believed that the sugar chains of PD-1 have no physical contact with Pembrolizumab due to the N-linked glycosylated residues (Asn49, Asn58, Asn74, and Asn116) being located away from the interface <ref name="horita" />. | The Fv fragment of Pembrolizumab can form a complex with the extracellular domain (ECD) of PD-1. Both PemFv and PD-1<sub>ECD</sub> contain interchain disulfide bonds. PemFv interacts predominantly in the major groove of PD-1, which is formed on one surface by the CC’FG antiparallel β−sheet and the BC, C’D, and FG loops. There are 15 direct hydrogen bonds between the residues, 15 water-mediated hydrogen bonds, 2 salt bridges, and many hydrophobic interactions. There are a total of 26 PD-1<sub>ECD</sub> residues involved in the interaction with PemFv, with residues in loop C’D (Pro84 to Gly90) and strand C’ (Gln75 to Lys 78) playing a major role. These key components of PD-1 mainly form interactions through salt bridges and hydrogen bonds with complementary determining regions, the variable domains, of Pembrolizumab. <scene name='74/745945/Chain_b_amino_acids/1'>Thr30, Tyr33, Ser54, Tyr101, Arg102</scene> on Chain B of Pembrolizumab form bonds with Asp77, Gln75, Lys78, Thr76, Tyr68, and Asn66 of PD-1. It is believed that the sugar chains of PD-1 have no physical contact with Pembrolizumab due to the N-linked glycosylated residues (Asn49, Asn58, Asn74, and Asn116) being located away from the interface <ref name="horita" />. | ||
Revision as of 02:53, 6 December 2016
Pembrolizumab antibody against programmed cell death-1 receptor
| |||||||||||
References
- ↑ 1.0 1.1 Longoria TC, Tewari KS. Evaluation of the pharmacokinetics and metabolism of pembrolizumab in the treatment of melanoma. Expert Opin Drug Metab Toxicol. 2016 Oct;12(10):1247-53. doi:, 10.1080/17425255.2016.1216976. Epub 2016 Aug 16. PMID:27485741 doi:http://dx.doi.org/10.1080/17425255.2016.1216976
- ↑ 2.0 2.1 2.2 2.3 Horita S, Nomura Y, Sato Y, Shimamura T, Iwata S, Nomura N. High-resolution crystal structure of the therapeutic antibody pembrolizumab bound to the human PD-1. Sci Rep. 2016 Oct 13;6:35297. doi: 10.1038/srep35297. PMID:27734966 doi:http://dx.doi.org/10.1038/srep35297
- ↑ Rajakulendran T, Adam DN. Spotlight on pembrolizumab in the treatment of advanced melanoma. Drug Des Devel Ther. 2015 Jun 4;9:2883-6. doi: 10.2147/DDDT.S78036. eCollection, 2015. PMID:26082618 doi:http://dx.doi.org/10.2147/DDDT.S78036
- ↑ Deeks ED. Pembrolizumab: A Review in Advanced Melanoma. Drugs. 2016 Mar;76(3):375-86. doi: 10.1007/s40265-016-0543-x. PMID:26846323 doi:http://dx.doi.org/10.1007/s40265-016-0543-x