This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Finasteride
From Proteopedia
(Difference between revisions)
| Line 5: | Line 5: | ||
==Function== | ==Function== | ||
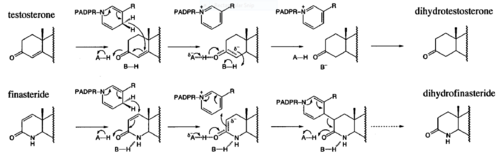
| - | Finasteride, branded as Proscar or Propecia, is a synthetic 4-azasteroid compound that acts as a 5α-reductase inhibitor.<ref name="one"> I.K. Morton; Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 121, 246. ISBN 978-94-011-4439-1 </ref> The 5α-reductase enzyme is very important in the metabolism of many of the steroids produced by the body, in particular the conversion of testosterone to dihydrotestosterone (DHT).<ref name="one"> I.K. Morton; Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 121, 246. ISBN 978-94-011-4439-1 </ref> For this reason, Finasteride is used as a treatment for benign prostate hyperplasia (BPH), which is caused by an overproduction of DHT in the male prostate.<ref name="two"> Yamana K, Labrie F, Luu-The V (January 2010). Human type 3 5α-reductase is expressed in peripheral tissues at higher levels than types 1 and 2 and its activity is potently inhibited by finasteride and dutasteride. Hormone Molecular Biology and Clinical Investigation. 2 (3). doi:10.1515/hmbci.2010.035 </ref> Androgenetic Alopecia or male pattern baldness (MPB) is another condition in men caused by the build up of DHT, which can also be treated with Finasteride.<ref name="three"> Varothai, S; Bergfeld, WF (Jul 2014). "Androgenetic alopecia: an evidence-based treatment update.". American journal of clinical dermatology. 15 (3): 217–30. doi:10.1007/s40257-014-0077-5. PMID 24848508 </ref> Finasteride's affinity for 5α-reductase approaches that of testosterone, and can bind to two of the three isoenzymes of 5α-reductase, types I and II.<ref name="two"> Yamana K, Labrie F, Luu-The V (January 2010). Human type 3 5α-reductase is expressed in peripheral tissues at higher levels than types 1 and 2 and its activity is potently inhibited by finasteride and dutasteride. Hormone Molecular Biology and Clinical Investigation. 2 (3). doi:10.1515/hmbci.2010.035 </ref> | + | '''Finasteride''', branded as Proscar or Propecia, is a synthetic 4-azasteroid compound that acts as a 5α-reductase inhibitor.<ref name="one"> I.K. Morton; Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 121, 246. ISBN 978-94-011-4439-1 </ref> The 5α-reductase enzyme is very important in the metabolism of many of the steroids produced by the body, in particular the conversion of testosterone to dihydrotestosterone (DHT).<ref name="one"> I.K. Morton; Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 121, 246. ISBN 978-94-011-4439-1 </ref> For this reason, Finasteride is used as a treatment for benign prostate hyperplasia (BPH), which is caused by an overproduction of DHT in the male prostate.<ref name="two"> Yamana K, Labrie F, Luu-The V (January 2010). Human type 3 5α-reductase is expressed in peripheral tissues at higher levels than types 1 and 2 and its activity is potently inhibited by finasteride and dutasteride. Hormone Molecular Biology and Clinical Investigation. 2 (3). doi:10.1515/hmbci.2010.035 </ref> Androgenetic Alopecia or male pattern baldness (MPB) is another condition in men caused by the build up of DHT, which can also be treated with Finasteride.<ref name="three"> Varothai, S; Bergfeld, WF (Jul 2014). "Androgenetic alopecia: an evidence-based treatment update.". American journal of clinical dermatology. 15 (3): 217–30. doi:10.1007/s40257-014-0077-5. PMID 24848508 </ref> Finasteride's affinity for 5α-reductase approaches that of testosterone, and can bind to two of the three isoenzymes of 5α-reductase, types I and II.<ref name="two"> Yamana K, Labrie F, Luu-The V (January 2010). Human type 3 5α-reductase is expressed in peripheral tissues at higher levels than types 1 and 2 and its activity is potently inhibited by finasteride and dutasteride. Hormone Molecular Biology and Clinical Investigation. 2 (3). doi:10.1515/hmbci.2010.035 </ref> |
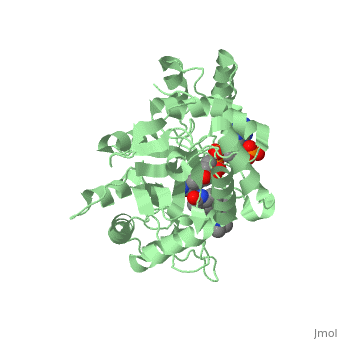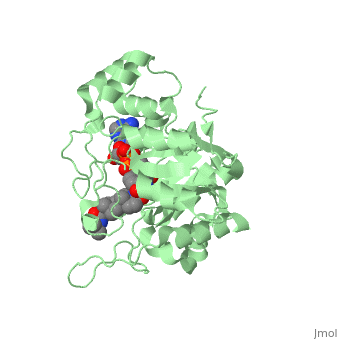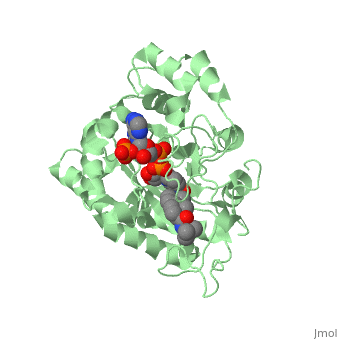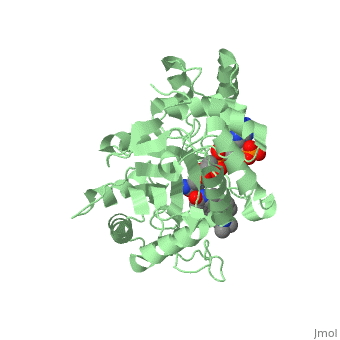
==Structure== | ==Structure== | ||
Revision as of 16:15, 16 January 2017
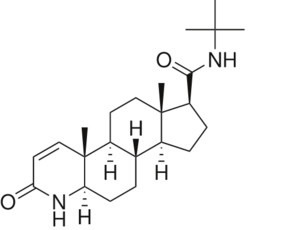
N-(1,1-dimethylethyl)-3-oxo-(5α,17β)-4-azaandrost-1-ene-17-carboxamide (Finasteride)
| |||||||||||
References
- ↑ 1.0 1.1 I.K. Morton; Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 121, 246. ISBN 978-94-011-4439-1
- ↑ 2.0 2.1 2.2 Yamana K, Labrie F, Luu-The V (January 2010). Human type 3 5α-reductase is expressed in peripheral tissues at higher levels than types 1 and 2 and its activity is potently inhibited by finasteride and dutasteride. Hormone Molecular Biology and Clinical Investigation. 2 (3). doi:10.1515/hmbci.2010.035
- ↑ Varothai, S; Bergfeld, WF (Jul 2014). "Androgenetic alopecia: an evidence-based treatment update.". American journal of clinical dermatology. 15 (3): 217–30. doi:10.1007/s40257-014-0077-5. PMID 24848508
- ↑ 4.0 4.1 4.2 Lednicer D (2011). Steroid Chemistry at a Glance. Hoboken: Wiley. ISBN 978-0-470-66084-3
- ↑ Burkhard Fugmann; Susanne Lang-Fugmann; Wolfgang Steglich (28 May 2014). RÖMPP Encyclopedia Natural Products, 1st Edition, 2000. Thieme. pp. 1918–. ISBN 978-3-13-179551-9
- ↑ Schieck, Cynthia L.(1998, August) "Finasteride (Propecia ®)". http://www.chm.bris.ac.uk/motm/finasteride/Finasteride%20(Propecia)%20-%20Feature%20Molecule.htm
- ↑ 7.0 7.1 Bull, Herbert G.*Garcia-Calvo,Margarita Andersson,Stefan†, Baginsky, Walter F.,Chan,H. Karen,Ellsworth,‡ Dina E., Miller,§ Randall R., Stearns,Ralph A.,Bakshi,Raman K.,Rasmusson, Gary H.,Tolman,Richard L., Myers,Robert W.,Kozarich,John W.,Harris,Georgianna S. (1995, August 6) Mechanism-Based Inhibition of Human Steroid 5R-Reductase by Finasteride: Enzyme-Catalyzed Formation of NADP-Dihydrofinasteride, a Potent Bisubstrate Analog Inhibitor. http://pubs.acs.org/doi/pdf/10.1021/ja953069t
- ↑ Tacklind, J., Fink, H.A., MacDonald, R., Rutks, I., Wilt, T.J. (2010). Finasteride for benign prostatic hyperplasia. Cochrane database of systematic reviews 2010, Issue 10. Art. No.: CD006015. DOI: 10.1002/14651858.CD006015.pub3.
- ↑ Dragan, I., Misso, M. (2012). Lycopene for the prevention and treatment of benign prostatic hyperplasia and prostate cancer: A systematic review. Maturitas, 72 (4), 269
- ↑ 10.0 10.1 Robaire, B., Henderson, N.A. (2006). Actions of 5alpha-reductase inhibitors on the epididymis. Molecular and Cellular Endocrinology. 250 (1-2): 190–5. doi:10.1016/j.mce.2005.12.044
- ↑ Paus, R., Cotsarelis, G. (1999).The biology of hair follicles. N. Engl. J. Med., 34, 491–497.
- ↑ Olsen, E. A., Hordinsky, M., & Whiting, D., et al. (2006, December).
- ↑ Leyden, James et al.(June 1999)."Finasteride in the treatment of men with frontal male pattern hair loss." Journal of the American Academy of Dermatology. Volume 40 , Issue 6 , 930 - 937
Proteopedia Page Contributors and Editors (what is this?)
Joel L. Sussman, Cody J Cubbage, Michal Harel, Alexander Berchansky