This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Estelle Metzger/Sandbox
From Proteopedia
(Difference between revisions)
| Line 16: | Line 16: | ||
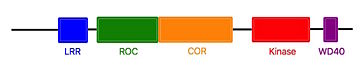
The COR and the ROC domains forms an inseperable tandem, a 300-400 long stretch of amino acids with no significant homology to other described domains. <ref name="Bernd2"/> | The COR and the ROC domains forms an inseperable tandem, a 300-400 long stretch of amino acids with no significant homology to other described domains. <ref name="Bernd2"/> | ||
| - | The Roco4 kinase structure consists of a canonical, two-lobed kinase structure, with an adenine nucleotide bound in the conventional nucleotide-binding pocket. It contains the conserved alphaC-helix and an anti-parallel beta sheets in the smaller N-terminal lobe. Other Alpha-helices and the activation loop with the conserved N-terminal DFG motif are localized in the bigger C-terminal lobe. | + | The Roco4 kinase structure consists of a canonical, two-lobed kinase structure, with an adenine nucleotide bound in the conventional nucleotide-binding pocket. It contains the conserved alphaC-helix and an anti-parallel beta sheets in the smaller N-terminal lobe. Other Alpha-helices and the activation loop with the conserved N-terminal DFG motif are localized in the bigger C-terminal lobe.<ref name="Bernd2"/> |
The activation loop and alphaC-helix together form the catalytic site of the kinase, an ATP binding site formed by a cleft between the two lobes. | The activation loop and alphaC-helix together form the catalytic site of the kinase, an ATP binding site formed by a cleft between the two lobes. | ||
For catalysis, the formation of a polar contact is essential. This polar contact takes place between Roco4 Lys1055 from the beta3-strand and the Glu1078 from the alphaC-helix. The amino acids Asp makes contact with all three ATP phosphates either directly or via coordination of a magnesium ion. Moreover, the amino acid Phe makes hydrophobic contacts to the alphaC-helix and the HxD motif, and leads for the correct positioning of the DFG motif. <ref name="Bernd2"/> | For catalysis, the formation of a polar contact is essential. This polar contact takes place between Roco4 Lys1055 from the beta3-strand and the Glu1078 from the alphaC-helix. The amino acids Asp makes contact with all three ATP phosphates either directly or via coordination of a magnesium ion. Moreover, the amino acid Phe makes hydrophobic contacts to the alphaC-helix and the HxD motif, and leads for the correct positioning of the DFG motif. <ref name="Bernd2"/> | ||
Revision as of 18:19, 26 January 2017
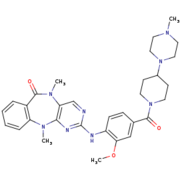
Humanized Roco4 bound to LRRK2-IN-1
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Gilsbach BK, Messias AC, Ito G, Sattler M, Alessi DR, Wittinghofer A, Kortholt A. Structural Characterization of LRRK2 Inhibitors. J Med Chem. 2015 May 1. PMID:25897865 doi:http://dx.doi.org/10.1021/jm5018779
- ↑ 2.0 2.1 2.2 2.3 2.4 Gilsbach BK, Kortholt A. Structural biology of the LRRK2 GTPase and kinase domains: implications for regulation. Front Mol Neurosci. 2014 May 5;7:32. doi: 10.3389/fnmol.2014.00032. eCollection, 2014. PMID:24847205 doi:http://dx.doi.org/10.3389/fnmol.2014.00032
- ↑ 3.0 3.1 doi: https://dx.doi.org/10.1016/S0092-8674(02)00741-9
- ↑ 4.0 4.1 Taylor SS, Kornev AP. Protein kinases: evolution of dynamic regulatory proteins. Trends Biochem Sci. 2011 Feb;36(2):65-77. doi: 10.1016/j.tibs.2010.09.006. Epub, 2010 Oct 23. PMID:20971646 doi:10.1016/j.tibs.2010.09.006