This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1s5o
From Proteopedia
| Line 5: | Line 5: | ||
|SITE= | |SITE= | ||
|LIGAND= <scene name='pdbligand=152:CARNITINE'>152</scene> | |LIGAND= <scene name='pdbligand=152:CARNITINE'>152</scene> | ||
| - | |ACTIVITY= [http://en.wikipedia.org/wiki/Carnitine_O-acetyltransferase Carnitine O-acetyltransferase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=2.3.1.7 2.3.1.7] | + | |ACTIVITY= <span class='plainlinks'>[http://en.wikipedia.org/wiki/Carnitine_O-acetyltransferase Carnitine O-acetyltransferase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=2.3.1.7 2.3.1.7] </span> |
|GENE= | |GENE= | ||
| + | |DOMAIN= | ||
| + | |RELATEDENTRY= | ||
| + | |RESOURCES=<span class='plainlinks'>[http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=1s5o FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1s5o OCA], [http://www.ebi.ac.uk/pdbsum/1s5o PDBsum], [http://www.rcsb.org/pdb/explore.do?structureId=1s5o RCSB]</span> | ||
}} | }} | ||
| Line 14: | Line 17: | ||
==Overview== | ==Overview== | ||
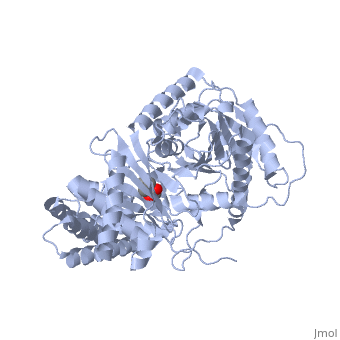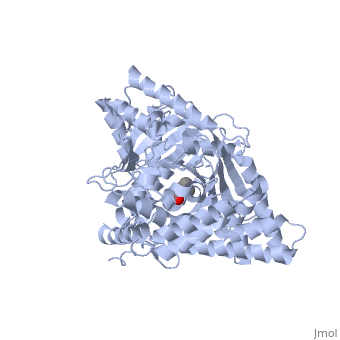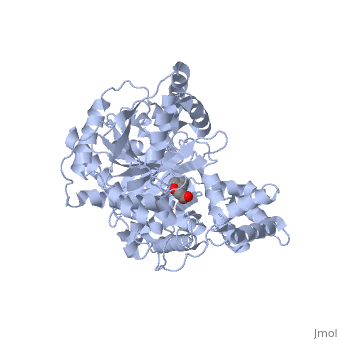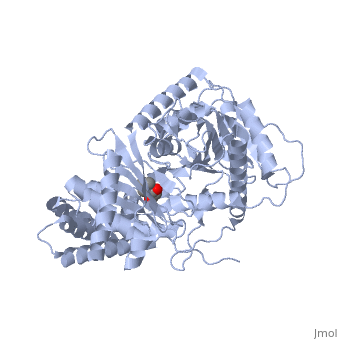
We report the crystal structure of a binary complex of human peroxisomal carnitine acetyltransferase and the substrate l-carnitine, refined to a resolution of 1.8 Angstrom with an R(factor) value of 18.9% (R(free)=22.3%). L-carnitine binds to a preformed pocket in the active site tunnel of carnitine acetyltransferase aligned with His(322). The quaternary nitrogen of carnitine forms a pi-cation interaction with Phe(545), while Arg(497) forms an electrostatic interaction with the negatively charged carboxylate group. An extensive hydrogen bond network also occurs between the carboxylate group and Tyr(431), Thr(444), and a bound water molecule. Site-directed mutagenesis and kinetic characterization reveals that Tyr(431), Thr(444), Arg(497), and Phe(545) are essential for high affinity binding of L-carnitine. | We report the crystal structure of a binary complex of human peroxisomal carnitine acetyltransferase and the substrate l-carnitine, refined to a resolution of 1.8 Angstrom with an R(factor) value of 18.9% (R(free)=22.3%). L-carnitine binds to a preformed pocket in the active site tunnel of carnitine acetyltransferase aligned with His(322). The quaternary nitrogen of carnitine forms a pi-cation interaction with Phe(545), while Arg(497) forms an electrostatic interaction with the negatively charged carboxylate group. An extensive hydrogen bond network also occurs between the carboxylate group and Tyr(431), Thr(444), and a bound water molecule. Site-directed mutagenesis and kinetic characterization reveals that Tyr(431), Thr(444), Arg(497), and Phe(545) are essential for high affinity binding of L-carnitine. | ||
| - | |||
| - | ==Disease== | ||
| - | Known disease associated with this structure: Carnitine acetyltransferase deficiency (1) OMIM:[[http://www.ncbi.nlm.nih.gov/entrez/dispomim.cgi?id=600184 600184]] | ||
==About this Structure== | ==About this Structure== | ||
| Line 35: | Line 35: | ||
[[Category: Pedersen, B.]] | [[Category: Pedersen, B.]] | ||
[[Category: Wu, D.]] | [[Category: Wu, D.]] | ||
| - | [[Category: 152]] | ||
[[Category: binary complex]] | [[Category: binary complex]] | ||
[[Category: carnitine acetyltransferase]] | [[Category: carnitine acetyltransferase]] | ||
| Line 42: | Line 41: | ||
[[Category: x-ray structure]] | [[Category: x-ray structure]] | ||
| - | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Sun Mar 30 23:37:44 2008'' |
Revision as of 20:37, 30 March 2008
| |||||||
| , resolution 1.8Å | |||||||
|---|---|---|---|---|---|---|---|
| Ligands: | |||||||
| Activity: | Carnitine O-acetyltransferase, with EC number 2.3.1.7 | ||||||
| Resources: | FirstGlance, OCA, PDBsum, RCSB | ||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||
Structural and Mutational Characterization of L-carnitine Binding to Human carnitine Acetyltransferase
Overview
We report the crystal structure of a binary complex of human peroxisomal carnitine acetyltransferase and the substrate l-carnitine, refined to a resolution of 1.8 Angstrom with an R(factor) value of 18.9% (R(free)=22.3%). L-carnitine binds to a preformed pocket in the active site tunnel of carnitine acetyltransferase aligned with His(322). The quaternary nitrogen of carnitine forms a pi-cation interaction with Phe(545), while Arg(497) forms an electrostatic interaction with the negatively charged carboxylate group. An extensive hydrogen bond network also occurs between the carboxylate group and Tyr(431), Thr(444), and a bound water molecule. Site-directed mutagenesis and kinetic characterization reveals that Tyr(431), Thr(444), Arg(497), and Phe(545) are essential for high affinity binding of L-carnitine.
About this Structure
1S5O is a Single protein structure of sequence from Homo sapiens. Full crystallographic information is available from OCA.
Reference
Structural and mutational characterization of L-carnitine binding to human carnitine acetyltransferase., Govindasamy L, Kukar T, Lian W, Pedersen B, Gu Y, Agbandje-McKenna M, Jin S, McKenna R, Wu D, J Struct Biol. 2004 Jun;146(3):416-24. PMID:15099582
Page seeded by OCA on Sun Mar 30 23:37:44 2008
Categories: Carnitine O-acetyltransferase | Homo sapiens | Single protein | Agbandje-Mckenna, M. | Govindasamy, L. | Gu, Y. | Jin, S. | Kukar, T. | Lian, W. | Mckenna, R. | Pedersen, B. | Wu, D. | Binary complex | Carnitine acetyltransferase | Steady-state enzyme kinetic | Substrate binding site | X-ray structure