This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1051
From Proteopedia
(Difference between revisions)
| Line 8: | Line 8: | ||
== Structural Overview == | == Structural Overview == | ||
| + | CzrA functions as a dimer. The <scene name='69/694218/Monomeric_unit/1'>monomeric units</scene> dimerize at the czr operon, repressing gene transcription. Each monomeric unit contains <scene name='69/694218/Helices/1'>five alpha helices</scene> seen in purple and <scene name='69/694218/B_sheets/1'>two beta sheets</scene> displayed in yellow. While the function of the beta sheets are not yet known, key helices regulate the binding of DNA and Zn<sup> +2 </sup>. The <scene name='69/694218/Alpha_4_helix/1'>alpha 4 helix</scene> is the location of DNA binding and the <scene name='69/694218/Alpha_5_helix/1'>alpha 5 helix</scene> contains the Zn<sup> +2 </sup> binding site. As Zn<sup> +2 </sup> bind, repressing their DNA binding ability. | ||
== Binding of DNA == | == Binding of DNA == | ||
The <scene name='69/694219/Serandhisresidues/1'>main DNA binding residues</scene> have been found to be the SER 54 and 57 along with HIS 58. | The <scene name='69/694219/Serandhisresidues/1'>main DNA binding residues</scene> have been found to be the SER 54 and 57 along with HIS 58. | ||
| + | |||
== Zinc Binding == | == Zinc Binding == | ||
| - | Zinc is an allosteric inhibitor to CzrA. Two | + | Zinc is an allosteric inhibitor to CzrA. Two Zn<sup> +2 </sup> ions may bind to the dimer, at the location of the <scene name='69/694218/Alpha_5_helices/2'> alpha 5 </scene> helix from each monomer. As zinc binds, the alpha 5 helices <scene name='69/694218/2kjc_zinc_bound/1'>swing down</scene> to inhibit the DNA binding residues. Furthermore, CzrA must be in its dimer form for zinc to bind. The <scene name='69/694218/Spacefill_with_zinc_pockets/1'>zinc binding pocket</scene> is formed by two residues from each monomer, so Zn<sup>+2</sup> cannot bind to the monomer. The <scene name='69/694218/Zinc_residues/1'>zinc binding site</scene> is formed by Asp84 and His86 from one monomer, and His97 and His100 from the other monomer. |
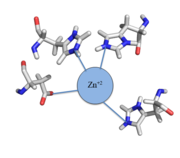
| - | The zinc<sup>+2</sup> ion forms a tetrahedral complex with the four residues (Figure 1). This allows other metal ions to act as allosteric inhibitors to CzrA. Any metal that may form a tetrahedral complex will have some affinity for CzrA, assuming it is not too large to fit into the pocket. However, the metal binding pocket of CzrA has been optimized to bind | + | The zinc<sup>+2</sup> ion forms a tetrahedral complex with the four residues (Figure 1). This allows other metal ions to act as allosteric inhibitors to CzrA. Any metal that may form a tetrahedral complex will have some affinity for CzrA, assuming it is not too large to fit into the pocket. However, the metal binding pocket of CzrA has been optimized to bind Zn<sup>+2</sup> with the highest affinity. As CzrA is a transcriptional repressor, binding of Zn<sup>+2</sup> to the dimer will activate the czr operon. Zn<sup>+2</sup> is preferred as CzrB opens a Zn<sup>+2</sup> channel, allowing the excess zinc ions to export the cell. |
[[Image:Zinc tetrahedral complex.PNG|thumb|center| Figure 1:Zn<sup>+2</sup> tetrahedral binding complex]] | [[Image:Zinc tetrahedral complex.PNG|thumb|center| Figure 1:Zn<sup>+2</sup> tetrahedral binding complex]] | ||
== References == | == References == | ||
<references/> | <references/> | ||
Revision as of 12:49, 28 March 2017
| This Sandbox is Reserved from 02/09/2015, through 05/31/2016 for use in the course "CH462: Biochemistry 2" taught by Geoffrey C. Hoops at the Butler University. This reservation includes Sandbox Reserved 1051 through Sandbox Reserved 1080. |
To get started:
More help: Help:Editing |
CzrA
| |||||||||||