This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1069
From Proteopedia
| Line 38: | Line 38: | ||
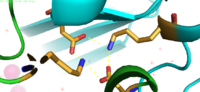
When YiiP is saturated with Zn<sup>2+</sup> it seems to favor the <scene name='69/694233/Outward-facing_conformation/2'>outward-facing conformation</scene> whereas when active sites are either empty or bound to H<sup>+</sup> the <scene name='69/694233/Outward-facing_conformation/1'>inward-facing conformation</scene> is favored. This drives the export of Zn<sup>2+</sup> from the cytoplasm and enhances the coupling of the proton-motive force. Although YiiP exists as a homodimer both monomers can undergo conformation change independent of one other to | When YiiP is saturated with Zn<sup>2+</sup> it seems to favor the <scene name='69/694233/Outward-facing_conformation/2'>outward-facing conformation</scene> whereas when active sites are either empty or bound to H<sup>+</sup> the <scene name='69/694233/Outward-facing_conformation/1'>inward-facing conformation</scene> is favored. This drives the export of Zn<sup>2+</sup> from the cytoplasm and enhances the coupling of the proton-motive force. Although YiiP exists as a homodimer both monomers can undergo conformation change independent of one other to | ||
produce the alternating access mechanism. | produce the alternating access mechanism. | ||
| - | |||
| - | == Electrostatic Interactions == | ||
| - | [[Image:Yiip_Electrostatic.png|250px|left|thumb|Electrostatic Charge Distribution]]<scene name='69/694234/Electro/1'>Charge distribution</scene> along the exterior surface of the protein is primarily neutral for the TMDs, but transitions to positive near the location of the charge interlock and interior side of the cell membrane. This positive section is characteristic of trans-membrane proteins as a means of achieving proper orientation within the cell membrane. Binding sites A, B, and C, as well as the CTDs of both monomers, all possess a high negative charge relative to the other charges present, facilitating the binding and releasing of Zn<sup>2+</sup> ions. The two CTDs are held together by the charge interlock and hydrophobic interactions of the TMDs, despite their electrostatic repulsion. Upon the release of Zn<sup>2+</sup> ions, the CTDs undergo alterations to electronegativity, which enables domain separation. | ||
===Zn<sup>2+</sup> Induced Conformation Change=== | ===Zn<sup>2+</sup> Induced Conformation Change=== | ||
| Line 47: | Line 44: | ||
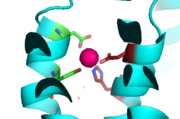
In contrast the main purpose of conformation change in the CTD is to stabilize the YiiP dimer and acts as a Zn<sup>2+</sup> sensor. This is possible because of the flexible loop that links the TMD and the CTD. This loop harbors the charge interlock which serves as a hinge that allows movement of the CTD. Using [https://en.wikipedia.org/wiki/F%C3%B6rster_resonance_energy_transfer FRET] to measure the distance between the CTD of each monomer fluorescence quenching was observed as the concentration Zn<sup>2+</sup> increased, which supports that idea that Zn<sup>2+</sup> induces a stabilizing conformation change in the CTD. | In contrast the main purpose of conformation change in the CTD is to stabilize the YiiP dimer and acts as a Zn<sup>2+</sup> sensor. This is possible because of the flexible loop that links the TMD and the CTD. This loop harbors the charge interlock which serves as a hinge that allows movement of the CTD. Using [https://en.wikipedia.org/wiki/F%C3%B6rster_resonance_energy_transfer FRET] to measure the distance between the CTD of each monomer fluorescence quenching was observed as the concentration Zn<sup>2+</sup> increased, which supports that idea that Zn<sup>2+</sup> induces a stabilizing conformation change in the CTD. | ||
| + | |||
| + | == Electrostatic Interactions == | ||
| + | [[Image:Yiip_Electrostatic.png|250px|left|thumb|Electrostatic Charge Distribution]]<scene name='69/694234/Electro/1'>Charge distribution</scene> along the exterior surface of the protein is primarily neutral for the TMDs, but transitions to positive near the location of the charge interlock and interior side of the cell membrane. This positive section is characteristic of trans-membrane proteins as a means of achieving proper orientation within the cell membrane. Binding sites A, B, and C, as well as the CTDs of both monomers, all possess a high negative charge relative to the other charges present, facilitating the binding and releasing of Zn<sup>2+</sup> ions. The two CTDs are held together by the charge interlock and hydrophobic interactions of the TMDs, despite their electrostatic repulsion. Upon the release of Zn<sup>2+</sup> ions, the CTDs undergo alterations to electronegativity, which enables domain separation. | ||
== References == | == References == | ||
<references/> | <references/> | ||
Revision as of 21:49, 29 March 2017
Introduction
Zinc transporter is an integral membrane protein found in the membrane of Esherichia coli and a member of the cation diffusion facilitator family. Members of this family occur all throughout the biological real, their primary function being the export of divalent transition metal ions from the cytoplasm to the extracellular space [1]. They work to regulate the amount of divalent metals inside of the cell, which is biologically relevant because while these metals are necessary for different biological functions, they can prove fatal to the cell in excess amounts. Zinc is essential for the growth and development of cells and zinc levels can affect everything from gene expression to immune response. While YiiP is an integral membrane protein in the cells of Escherichia coli, understanding the mechanism of regulation behind it can help researcher's better understand the cation diffusion facilitator equivalents in eukaryotic cells.
| |||||||||||