Caspase-6 is an endopeptidase involved in apoptosis. In terms of its catalytic function, it is a part of the cysteine-aspartate family. Before Caspase-6 becomes functional, the enzyme exists as a , also known as a zymogen. In its , the zymogen exists as a homodimer, whose are then cleaved at to assume its active conformation. Zymogen activation through cleavage is largely conserved across the caspase family. However, Caspase-6 is unique in that it becomes active through self-cleavage in addition to cleavage by a separate enzyme. Each monomeric unit of zymogen contains a consisting of two helices, a consisting of three helices, a , and a . After cleavage at all sites, the processed post-zymogen monomers remain closely associated together through intermolecular forces as a dimer.
Zymogen
In addition to a self-cleavage mechanism, Caspase-6 can be activated getting cleaved by Caspase-3, as well as other enzymes. The mechanism of activation by clevage is highly conserved across the caspase family; Self-processing is uniquely recognized as the primary mechanism for Caspase-6 activation, where clevage must occur at for complete activation, specifically the and the must be removed. These cleavages are both sequence specific and ordered, starting with cleavage of the pro-domain at . Then removal of the intersubunit linker occurs through cleavage at two sites, . It has been proposed that this sequence of cleavage is due to the pro-domain being more readily available to enter the active site, whose presence inhibits Caspase-6's ability to cleave the intersubunit loop and self-activate; The prodomain acts as a “suicide protector”, preventing the TEVD193 cleavage site from the active site[3]. After both cleavages occur, remains in solution as a dimer.
Active State

Substrate binding groove in Caspase-6. Blue - catalytic residues yellow - ligand red - generic surface
In its active state, caspase-6 utilizes a catalytic triad
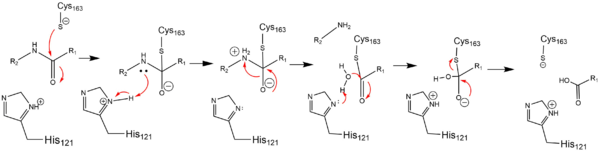
In order to function as an endoprotease, Caspase-6 utilzes a catalytic triad that binds a , which can include neuronal proteins and tubulins, in its active site. This binding groove contains three critical amino acid residues necessary to perform cleavage of the peptide bonds. A , composed of , , and , carries out cleavage of the substrate. In the theorized mechanism, His-121 acts as an acid catalyst, Glu-123 acts as a base catalyst to deprotonate Cys-163, which then acts as covalent catalyst.
Zinc Inhibition
Caspase-6 function is primarily inhibited by the binding of a ion, which binds to an instead of the . This allosteric site is located on the outside of the protein and is distal to the active site. The zinc ion is bound to , Lys-36, Glu-244, and His-287. Once the ion is bound to the protein, it is then stabilized by a found in the cytoplasm. The binding of zinc at the exosite is suggested to cause a conformational change in the protein from an to an that misaligns catalytic residues and inhibits activity of the enzyme. It has been proposed that helices of the active dimer must rotate or move in some other way to provide these ideal interactions with zinc. This subtle shift is most likely the cause for allosteric inhibition. As the helices move to bind zinc, the amino acids of the active site become misaligned. The altered positions of the amino acids no longer provide ideal interactions for incoming substrates. After zinc binds, no new substrates enter the active site. Thus, Caspase-6 is effectively inhibited. The residues in the active site no longer provide ideal interactions with the substrate and therefore, substrate does not bind. Zinc binding to the exosite is tightly regulated as it inhibits Caspase-6's critical role in initiation of apoptosis.
Phosphorylation
The function of Caspase-6 can be inhibited by phosphorylation of Ser-257. The exact mechanism of this reaction remains unidentified at the time of publication, but proceeds when ARK5 kinase is present. This modification can occur before and after zymogen activation. The phosphoryl group inhibits Caspase-6 through steric interference. When Ser-257 is phosphorylated, the amino acid residue interacts with , causing a shift in the helices of Caspase-6. This is shown in the , whose mutation mimics phosphorylation. [1] The shift misaligns and disrupts residues found in the active site. This conformational difference prevents the intersubunit linker from entering during zymogen activation and the self-cleaved active dimer cannot be formed. Additionally, no new substrate is able to enter the active site.
Medical Relevance
Caspase-6 involvement in Alzheimer's Disease
Caspase-6 is known to be involved in many neurodegenerative diseases, one of which is Alzheimer's disease (AD). Caspase-6 activity is associated with the formation of lesions within the Alzheimer's Disease.Lesions can be found in early stages of AD[2]. A proapoptotic protein, p53, is present at increased levels within AD brains, which seems to directly increase the transcription of Caspase-6, which indirectly influences apoptosis of neurons. Future treatments of AD include selective inhibition of active Caspase-6 proteins; staining has found active Caspase-6 within the hippocampus and cortex of the brain within a varying severity of AD cases. This suggests that Caspase-6 plays a predominate role in the pathophysiology of AD. There has been research conducted that shows activation of Caspase-6 in AD could cause disruption of the cytoskeleton network of neurons and lead to neuronal apoptosis[2].
Miscellaneous
Activation of Caspase-6
Before Caspase-6 is a functional and active dimer, the enzyme exists as a , also known as zymogen [1]. Caspase-6 can be activated by acting as a substrate for other caspases, particularly Caspase-3, as well as other enzymes. It becomes cleaved by these enzymes and proceeds to its . It was observed that Caspase-6 became active without alternate enzymes present, which suggested that Caspase-6 utilizes a self-cleavage mechanism. Now, self-processing, a characteristic unique to Caspase-6, is recognized as the primary mechanism for Caspase-6 activation. The unprocessed enzyme contains a and subunit, a , as well as an intersubunit linker. To become active, the intersubunit linker binds to the active site, where it is then cleaved. Other cleavages must occur as well for the enzyme to become active, specifically at TETD23 (these residues are not visible in the crystallized structure) of the pro-domain, , and amino acid sequences. Cleavage at these sites occurs in a . First, the site within the pro-domain, TETD23, must be cleaved. This cleavage is then followed by either DVVD179 or TEVD193.
Despite the sequence similarities between TETD23 and TEVD193 cleavage sites, the TETD23 cleavage site is always cleaved before TEVD193. It has been proposed that this sequence of cleavage is due to the , which allows the pro-domain to be more readily available to enter the active site. To some extent, the pro-domain inhibits Caspase-6's ability to cleave the intersubunit loop and self-activate, but this happens in a currently unknown mechanism. The result of the TETD23 cleavage site priority is that the prodomain acts as a “suicide protector”, which protects the TEVD193 cleavage site from self-cleavage[3]. This protection is necessary when there are low levels of inactive proteins, which must be preserved, in the tissue. The pro-domain is released after the cleavage at TETD23 and cleavage of the intersubunit links follow. This then allows the two subunits to interact to form the active dimer. The intramolecular cleavage of TEVD193 is essential for the initiation caspase-6 activation without other caspases present.


