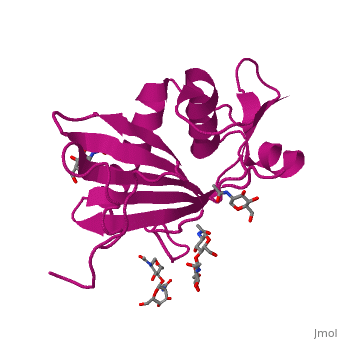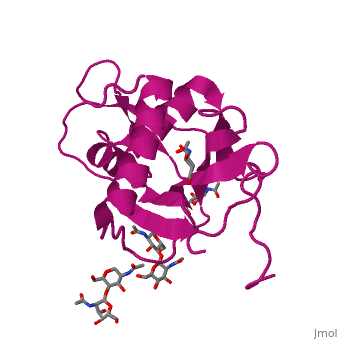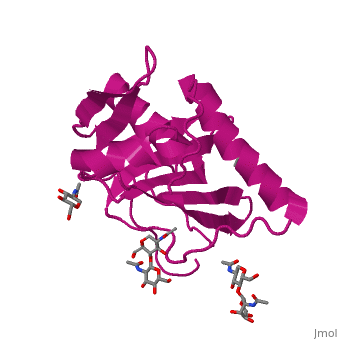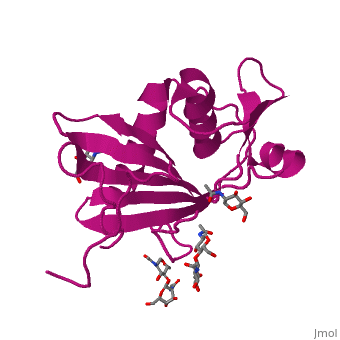This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
GP1 of Lassa Virus
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
<StructureSection load='4zjf' size='340' side='right' caption='4ZJF structure' scene='76/761695/4zjf_chaina/2'> | <StructureSection load='4zjf' size='340' side='right' caption='4ZJF structure' scene='76/761695/4zjf_chaina/2'> | ||
==Importance== | ==Importance== | ||
| - | Lassa virus (LASV), an Old World [http://en.wikipedia.org/wiki/Arenavirus arenavirus], is a notorious disease-causing agent primarily in West Africa that is able to spread to rodents, as well as humans. This deadly pathogen causes severe viral hemorrhagic fevers and significant mortality. So far, there are no available vaccines for LASV or any other viruses found in the ''Arenaviridae'' family. Determining the structure of the complete trimeric glycoprotein complex (GPC), composed of GP1, GP2, and SSP (stable signal peptide), will lay the foundation for a future discovery of novel antiviral drugs. This is the first representative structure for Old World arenaviruses. | + | Lassa virus (LASV), an Old World [http://en.wikipedia.org/wiki/Arenavirus arenavirus], is a notorious disease-causing agent primarily in West Africa that is able to spread to rodents, as well as to humans. This deadly pathogen causes severe viral hemorrhagic fevers and significant mortality. So far, there are no available vaccines for LASV or any other viruses found in the ''Arenaviridae'' family. Structural data at atomic resolution for viral proteins is key for understanding their function at the molecular level and can facilitate novel avenues for combating viral infections. Determining the structure of the complete trimeric glycoprotein complex (GPC), composed of GP1, GP2, and SSP (stable signal peptide), will lay the foundation for a future discovery of novel antiviral drugs. This is the first representative structure for Old World arenaviruses. This structure reveals the overall architecture of GP1 domains from Old World arenaviruses. From this structure came important information relating to the mechanisms for pH switching and the binding of Lassa virus to LAMP1, a recently identified host receptor that is critical for successful infection. In addition, structural analysis suggests two novel immune evasion mechanisms that Lassa virus may utilize to escape antibody-based immune response. |
== Function == | == Function == | ||
'''GP1''' (Glycoprotein 1) is the receptor binding domain of LASV that mediates receptor recognition. Research thus far indicates that GP1 from LASV may undergo irreversible conformational changes that could serve as an immunological decoy mechanism. | '''GP1''' (Glycoprotein 1) is the receptor binding domain of LASV that mediates receptor recognition. Research thus far indicates that GP1 from LASV may undergo irreversible conformational changes that could serve as an immunological decoy mechanism. | ||
Revision as of 10:45, 6 July 2017
| |||||||||||
References
- ↑ 1.0 1.1 Cohen-Dvashi H, Cohen N, Israeli H, Diskin R. Molecular mechanism for LAMP1 recognition by Lassa Virus. J Virol. 2015 May 13. pii: JVI.00651-15. PMID:25972533 doi:http://dx.doi.org/10.1128/JVI.00651-15
- ↑ Cao W, Henry MD, Borrow P, Yamada H, Elder JH, Ravkov EV, Nichol ST, Compans RW, Campbell KP, Oldstone MB. Identification of alpha-dystroglycan as a receptor for lymphocytic choriomeningitis virus and Lassa fever virus. Science. 1998 Dec 11;282(5396):2079-81. PMID:9851928
- ↑ Kunz S, Rojek JM, Perez M, Spiropoulou CF, Oldstone MB. Characterization of the interaction of lassa fever virus with its cellular receptor alpha-dystroglycan. J Virol. 2005 May;79(10):5979-87. PMID:15857984 doi:http://dx.doi.org/10.1128/JVI.79.10.5979-5987.2005
- ↑ 4.0 4.1 Eschli B, Quirin K, Wepf A, Weber J, Zinkernagel R, Hengartner H. Identification of an N-terminal trimeric coiled-coil core within arenavirus glycoprotein 2 permits assignment to class I viral fusion proteins. J Virol. 2006 Jun;80(12):5897-907. PMID:16731928 doi:http://dx.doi.org/10.1128/JVI.00008-06
- ↑ Li S, Sun Z, Pryce R, Parsy ML, Fehling SK, Schlie K, Siebert CA, Garten W, Bowden TA, Strecker T, Huiskonen JT. Acidic pH-Induced Conformations and LAMP1 Binding of the Lassa Virus Glycoprotein Spike. PLoS Pathog. 2016 Feb 5;12(2):e1005418. doi: 10.1371/journal.ppat.1005418., eCollection 2016 Feb. PMID:26849049 doi:http://dx.doi.org/10.1371/journal.ppat.1005418
- ↑ Oppliger J, Torriani G, Herrador A, Kunz S. Lassa Virus Cell Entry via Dystroglycan Involves an Unusual Pathway of Macropinocytosis. J Virol. 2016 Jun 24;90(14):6412-29. doi: 10.1128/JVI.00257-16. Print 2016 Jul, 15. PMID:27147735 doi:http://dx.doi.org/10.1128/JVI.00257-16
Categories: Cohen, N | Cohen-Dvashi, H | Diskin, R | Israeli, H | Arenavirus | Glycoprotein | Lassa | LASV | Receptor binding | Viral protein | 4zjf | GP1