This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
2-isopropylmalate synthase
From Proteopedia
(Difference between revisions)
| Line 6: | Line 6: | ||
== Structural highlights == | == Structural highlights == | ||
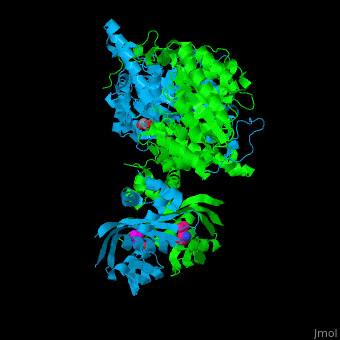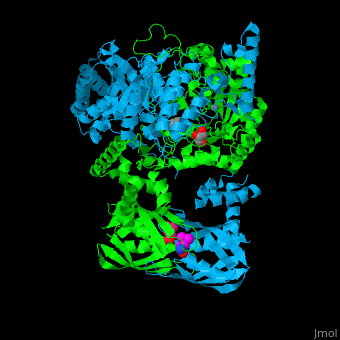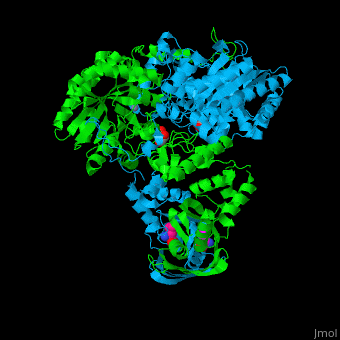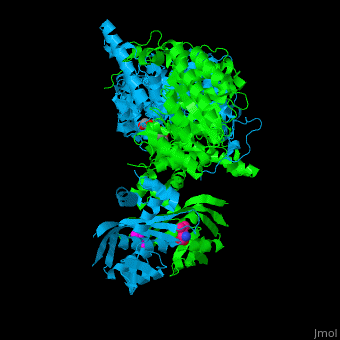
| - | <scene name='74/748866/Cv/2'>Domain organization of LeuA</scene>. LeuA structure is composed of 2 major domains - the catalytic N-terminal which shows a TIM barrel conformation and the regulatory C-terminal. The 2 domains are separated by subdomains I and II and a short flexible hinge region between the 2 subdomains. The catalytic domain binds the substrate and the <scene name='74/748866/Cv/3'>Zn+2 ion</scene><ref>PMID:15159544</ref>. | + | <scene name='74/748866/Cv/2'>Domain organization of LeuA</scene>. LeuA structure is composed of 2 major domains - the catalytic N-terminal which shows a TIM barrel conformation and the regulatory C-terminal. The 2 domains are separated by subdomains I and II and a short flexible hinge region between the 2 subdomains. The catalytic domain binds the substrate and the <scene name='74/748866/Cv/3'>Zn+2 ion</scene><ref>PMID:15159544</ref>. <scene name='74/748866/Cv/4'>Leucine binding site</scene>. |
</StructureSection> | </StructureSection> | ||
==3D structures of 2-isopropylmalate synthase== | ==3D structures of 2-isopropylmalate synthase== | ||
Revision as of 08:39, 3 August 2017
| |||||||||||
3D structures of 2-isopropylmalate synthase
Updated on 03-August-2017
References
- ↑ de Carvalho LP, Blanchard JS. Kinetic and chemical mechanism of alpha-isopropylmalate synthase from Mycobacterium tuberculosis. Biochemistry. 2006 Jul 25;45(29):8988-99. PMID:16846242 doi:http://dx.doi.org/10.1021/bi0606602
- ↑ Koon N, Squire CJ, Baker EN. Crystal structure of LeuA from Mycobacterium tuberculosis, a key enzyme in leucine biosynthesis. Proc Natl Acad Sci U S A. 2004 Jun 1;101(22):8295-300. Epub 2004 May 24. PMID:15159544 doi:10.1073/pnas.0400820101