We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
Polyamine oxidase
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
| - | + | <StructureSection load='3l1r' size='400' side='right' scene='1m10/Surface/2' caption='lycosylated FAD containing polyamine oxidase dimer complex with spermidine, sulfate and Cl- ion (green), [[3l1r]]' > | |
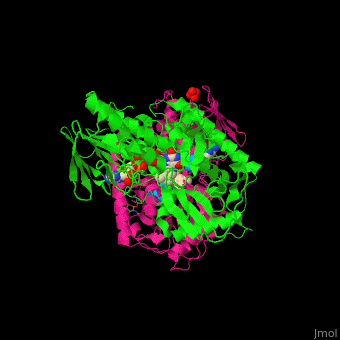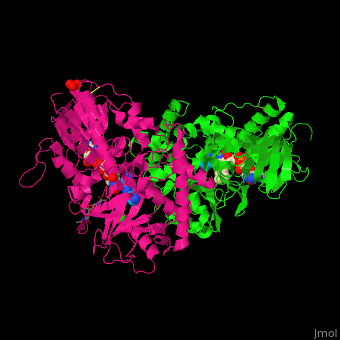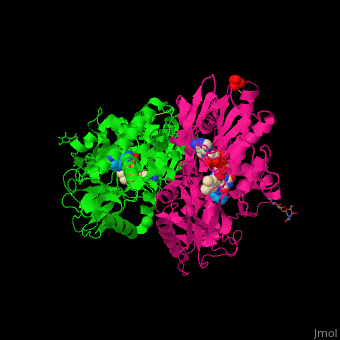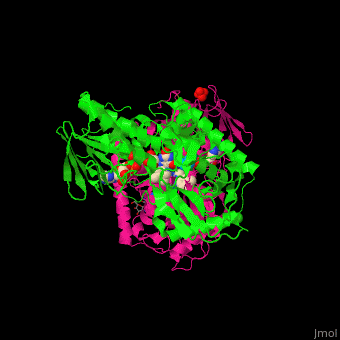
| - | '''Polyamine oxidase''' (PAO) catalyzes the conversion of N-acetylspermine, molecular oxygen and water to N-acetylspermidine, 3-aminopropanol and hydrogen peroxide. PAO via its production of hydrogen peroxide, is one of the key elements for oxidative burst which induces programmed cell death. PAO is involved in polyamine catabolism and uses FAD as a cofactor<ref>PMID:8584670</ref>. | ||
| + | '''Polyamine oxidase''' (PAO) catalyzes the conversion of N-acetylspermine, molecular oxygen and water to N-acetylspermidine, 3-aminopropanol and hydrogen peroxide. PAO via its production of hydrogen peroxide, is one of the key elements for oxidative burst which induces programmed cell death. PAO is involved in polyamine catabolism and uses FAD as a cofactor<ref>PMID:8584670</ref>. | ||
| + | </StructureSection> | ||
==3D structures of polyamine oxidase== | ==3D structures of polyamine oxidase== | ||
Revision as of 12:06, 7 September 2017
| |||||||||||
3D structures of polyamine oxidase
Updated on 07-September-2017
References
- ↑ Seiler N. Polyamine oxidase, properties and functions. Prog Brain Res. 1995;106:333-44. PMID:8584670