This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Ribonucleotide reductase
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
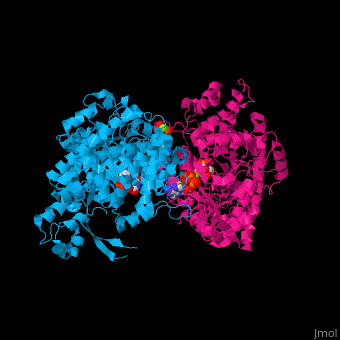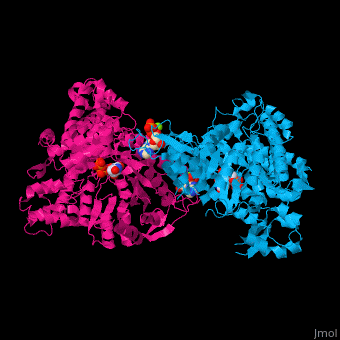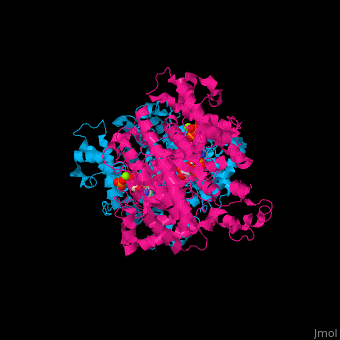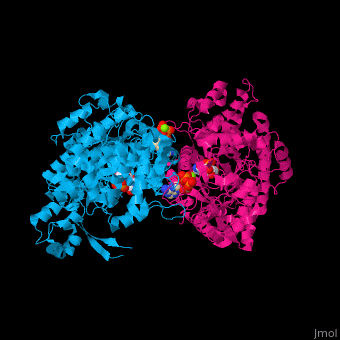
<StructureSection load='1xjg' size='450' side='right' caption='Class II ribonucleotide reductase dimer complex with dATP, UDP and Mg+2 ion (green) (PDB entry [[1xjg]])' scene='46/461381/Cv/1'> | <StructureSection load='1xjg' size='450' side='right' caption='Class II ribonucleotide reductase dimer complex with dATP, UDP and Mg+2 ion (green) (PDB entry [[1xjg]])' scene='46/461381/Cv/1'> | ||
== Function == | == Function == | ||
| - | '''Ribonucleotide reductase''' (RNR) catalyzes the formation of deoxyribonucleotides from ribonucleotides<ref>PMID:16756507</ref>. There are 3 classes of RNR.<br /> | + | '''Ribonucleotide reductase''' (RNR) or '''ribonucleotide-diphosphate reductase''' catalyzes the formation of deoxyribonucleotides from ribonucleotides<ref>PMID:16756507</ref>. There are 3 classes of RNR.<br /> |
* '''Class I RNR''' is a tetramer composed from large (RNR1) and small (RNR2) subunits. Class I RNR is iron-dependent and produces tyrosyl radical. Thimidine triphosphate (TTP) is an effector in the reaction.<br /> | * '''Class I RNR''' is a tetramer composed from large (RNR1) and small (RNR2) subunits. Class I RNR is iron-dependent and produces tyrosyl radical. Thimidine triphosphate (TTP) is an effector in the reaction.<br /> | ||
* ''' Class II RNR''' reduces ribonucleotide triphosphates using coenzyme B12.<br /> | * ''' Class II RNR''' reduces ribonucleotide triphosphates using coenzyme B12.<br /> | ||
| Line 28: | Line 28: | ||
**[[1zyz]], [[1zzd]], [[2cvs]], [[2cvt]], [[2cvu]], [[2cvv]], [[2cvw]], [[2cvx]], [[2cvy]] - yRNR R1 – yeast<BR /> | **[[1zyz]], [[1zzd]], [[2cvs]], [[2cvt]], [[2cvu]], [[2cvv]], [[2cvw]], [[2cvx]], [[2cvy]] - yRNR R1 – yeast<BR /> | ||
| - | **[[ | + | **[[1r1r]], [[1rlr]], [[1mrr]] - EcRNR R1 – ''Escherichia coli''<br /> |
| + | **[[5r1r]], [[6r1r]], [[7r1r]] - EcRNR R1 (mutant)<br /> | ||
**[[4n82]] – SsRNR – ''Streptococcus sanguinis''<br /> | **[[4n82]] – SsRNR – ''Streptococcus sanguinis''<br /> | ||
| Line 56: | Line 57: | ||
*RNR small subunit | *RNR small subunit | ||
| - | **[[2xof]], [[3n37]], [[3n38]], [[2alx]], [[1piy]], [[1r65]], [[1mxr]], [[1jqc]], [[1jpr]], [[1av8]], [[1xik]], [[1rib]], [[4m1f]] – EcRNR R2 <BR /> | + | **[[2xof]], [[3n37]], [[3n38]], [[2alx]], [[1piy]], [[1r65]], [[1mxr]], [[1jqc]], [[1jpr]], [[1av8]], [[1xik]], [[1rib]], [[4m1f]], [[5ci4]], [[4xbw]], [[5dco]], [[5dcr]], [[5dcs]], [[5ekb]], [[4xb9]], [[4xbv]] – EcRNR R2 <BR /> |
| - | **[[1yfd]], [[1piz]], [[1pj0]], [[1pj1]], [[1pm2]], [[1pim]], [[1piu]], [[1biq]], [[2av8]], [[1pfr]] - EcRNR R2 (mutant) + Fe<BR /> | + | **[[1yfd]], [[1piz]], [[1pj0]], [[1pj1]], [[1pm2]], [[1pim]], [[1piu]], [[1biq]], [[2av8]], [[1pfr]], [[5ci0]], [[5ci1]], [[5ci2]], [[5ci3]] - EcRNR R2 (mutant) + Fe<BR /> |
**[[3hf1]], [[2vux]], [[2uw2]], [[4djn]] – hRNR M2<BR /> | **[[3hf1]], [[2vux]], [[2uw2]], [[4djn]] – hRNR M2<BR /> | ||
**[[3mjo]], [[3dhz]], [[1oqu]], [[1kgn]], [[1kgo]], [[1kgp]] – RNR R2 – ''Corynebacterium ammoniagenes''<BR /> | **[[3mjo]], [[3dhz]], [[1oqu]], [[1kgn]], [[1kgo]], [[1kgp]] – RNR R2 – ''Corynebacterium ammoniagenes''<BR /> | ||
Revision as of 22:53, 27 September 2017
| |||||||||||
3D Structures of Ribonucleotide reductase
Updated on 27-September-2017
References
- ↑ Nordlund P, Reichard P. Ribonucleotide reductases. Annu Rev Biochem. 2006;75:681-706. PMID:16756507 doi:http://dx.doi.org/10.1146/annurev.biochem.75.103004.142443
- ↑ Munro JB, Silva JC. Ribonucleotide reductase as a target to control apicomplexan diseases. Curr Issues Mol Biol. 2012;14(1):9-26. Epub 2011 Jul 26. PMID:21791713
- ↑ Larsson KM, Jordan A, Eliasson R, Reichard P, Logan DT, Nordlund P. Structural mechanism of allosteric substrate specificity regulation in a ribonucleotide reductase. Nat Struct Mol Biol. 2004 Nov;11(11):1142-9. Epub 2004 Oct 10. PMID:15475969 doi:http://dx.doi.org/10.1038/nsmb838
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Joel L. Sussman, Jaime Prilusky