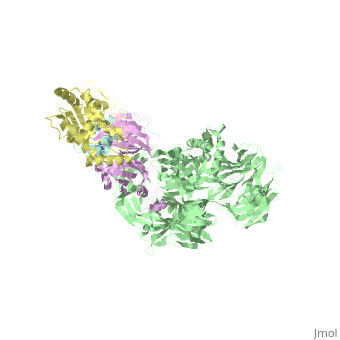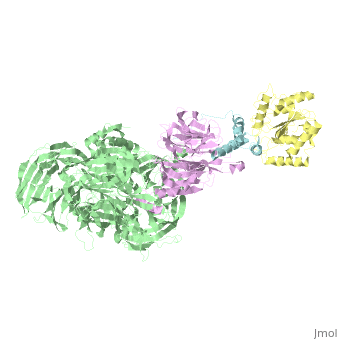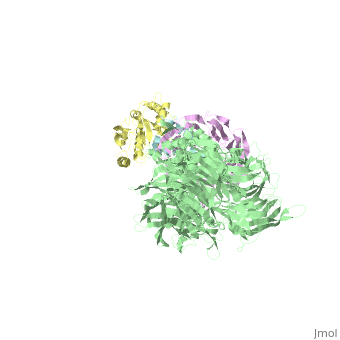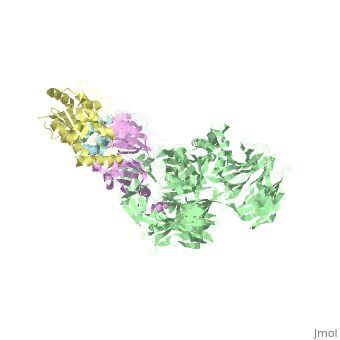
| Structural highlights
Disease
[UNG_HUMAN] Defects in UNG are a cause of immunodeficiency with hyper-IgM type 5 (HIGM5) [MIM:608106]. A rare immunodeficiency syndrome characterized by normal or elevated serum IgM levels with absence of IgG, IgA, and IgE. It results in a profound susceptibility to bacterial infections.[1] [2]
Function
[VPR_HV1N5] Involved in the transport of the viral pre-integration (PIC) complex to the nucleus during the early stages of the infection. This function is crucial for viral infection of non-dividing macrophages. May interact with karyopherin alpha/KPNA1 and KPNA2 to increase their affinity for proteins containing basic-type nuclear localization signal, including the viral matrix protein MA, thus facilitating the translocation of the viral genome into the nucleus. May also act directly at the nuclear pore complex, by binding nucleoporins phenylalanine-glycine (FG)-repeat regions (By similarity). May target specific host proteins for degradation by the 26S proteasome. Acts by associating with the cellular CUL4A-DDB1 E3 ligase complex through direct interaction with host VPRPB/DCAF-1. This change in the E3 ligase substrate specificity would result in cell cycle arrest or apoptosis in infected cells. Prevents infected cells from undergoing mitosis and proliferating, by inducing arrest or delay in the G2 phase of the cell cycle. This arrest creates a favorable environment for maximizing viral expression and production by rendering the HIV-1 LTR transcriptionally more active. In this context, Vpr stimulates gene expression driven by the HIV-1 LTR by interacting with human SP1, TFIIB and TFIID. Cell cycle arrest reportedly occurs within hours of infection and is not blocked by antiviral agents, suggesting that it is initiated by the Vpr carried into the virion. Additionally, Vpr induces apoptosis in a cell cycle dependent manner suggesting that these two effects are mechanistically linked. Interacts with mitochondrial permeability transition pore complex (PTPC). This interaction induces a rapid dissipation of the mitochondrial transmembrane potential, and mitochondrial release of apoptogenic proteins such as cytochrome C or apoptosis inducing factors. Detected in the serum and cerebrospinal fluid of AIDS patient, Vpr may also induce cell death to bystander cells (By similarity). [DDB1_HUMAN] Required for DNA repair. Binds to DDB2 to form the UV-damaged DNA-binding protein complex (the UV-DDB complex). The UV-DDB complex may recognize UV-induced DNA damage and recruit proteins of the nucleotide excision repair pathway (the NER pathway) to initiate DNA repair. The UV-DDB complex preferentially binds to cyclobutane pyrimidine dimers (CPD), 6-4 photoproducts (6-4 PP), apurinic sites and short mismatches. Also appears to function as a component of numerous distinct DCX (DDB1-CUL4-X-box) E3 ubiquitin-protein ligase complexes which mediate the ubiquitination and subsequent proteasomal degradation of target proteins. The functional specificity of the DCX E3 ubiquitin-protein ligase complex is determined by the variable substrate recognition component recruited by DDB1. DCX(DDB2) (also known as DDB1-CUL4-ROC1, CUL4-DDB-ROC1 and CUL4-DDB-RBX1) may ubiquitinate histone H2A, histone H3 and histone H4 at sites of UV-induced DNA damage. The ubiquitination of histones may facilitate their removal from the nucleosome and promote subsequent DNA repair. DCX(DDB2) also ubiquitinates XPC, which may enhance DNA-binding by XPC and promote NER. DCX(DTL) plays a role in PCNA-dependent polyubiquitination of CDT1 and MDM2-dependent ubiquitination of TP53 in response to radiation-induced DNA damage and during DNA replication. DCX(ERCC8) (the CSA complex) plays a role in transcription-coupled repair (TCR). May also play a role in ubiquitination of CDKN1B/p27kip when associated with CUL4 and SKP2.[3] [4] [5] [6] [7] [8] [9] [10] [11] [12] [13] [14] [15] [16] [17] [18] [UNG_HUMAN] Excises uracil residues from the DNA which can arise as a result of misincorporation of dUMP residues by DNA polymerase or due to deamination of cytosine. [VPRBP_HUMAN] Component of the CUL4A-RBX1-DDB1-VprBP/DCAF1 E3 ubiquitin-protein ligase complex, VprBP/DCAF1 may function as the substrate recognition module within this complex. For example, VprBP/DCAF1 targets NF2 to the E3 ubiquitin-ligase complex for ubiquitination and subsequent proteasome-dependent degradation. In case of infection by HIV-1 virus, it is recruited by HIV-1 Vpr in order to hijack the CUL4A-RBX1-DDB1 function leading to arrest the cell cycle in G2 phase, and also to protect the viral protein from proteasomal degradation by another E3 ubiquitin ligase. In case of infection by HIV-2 virus, it is recruited by HIV-2 Vpx in order to hijack the CUL4A-RBX1-DDB1 function leading to enhanced efficiency of macrophage infection and promotion of the replication of cognate primate lentiviruses in cells of monocyte/macrophage lineage. Associated with chromatin in a DDB1-independent and cell cycle-dependent manner, VprBP/DCAF1 is recruited to chromatin as DNA is being replicated and is released from chromatin before mitosis.[19] [20] [21] [22] [23] [24] [25] [26] [27] [28] [29] [30] [31]
Publication Abstract from PubMed
The HIV-1 accessory protein Vpr is required for efficient viral infection of macrophages and promotion of viral replication in T cells. Vpr's biological activities are closely linked to the interaction with human DCAF1, a cellular substrate receptor of the Cullin4-RING E3 ubiquitin ligase (CRL4) of the host ubiquitin-proteasome-mediated protein degradation pathway. The molecular details of how Vpr usurps the protein degradation pathway have not been delineated. Here we present the crystal structure of the DDB1-DCAF1-HIV-1-Vpr-uracil-DNA glycosylase (UNG2) complex. The structure reveals how Vpr engages with DCAF1, creating a binding interface for UNG2 recruitment in a manner distinct from the recruitment of SAMHD1 by Vpx proteins. Vpr and Vpx use similar N-terminal and helical regions to bind the substrate receptor, whereas different regions target the specific cellular substrates. Furthermore, Vpr uses molecular mimicry of DNA by a variable loop for specific recruitment of the UNG2 substrate.
The DDB1-DCAF1-Vpr-UNG2 crystal structure reveals how HIV-1 Vpr steers human UNG2 toward destruction.,Wu Y, Zhou X, Barnes CO, DeLucia M, Cohen AE, Gronenborn AM, Ahn J, Calero G Nat Struct Mol Biol. 2016 Aug 29. doi: 10.1038/nsmb.3284. PMID:27571178[32]
From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.
See Also
References
- ↑ Imai K, Slupphaug G, Lee WI, Revy P, Nonoyama S, Catalan N, Yel L, Forveille M, Kavli B, Krokan HE, Ochs HD, Fischer A, Durandy A. Human uracil-DNA glycosylase deficiency associated with profoundly impaired immunoglobulin class-switch recombination. Nat Immunol. 2003 Oct;4(10):1023-8. Epub 2003 Sep 7. PMID:12958596 doi:http://dx.doi.org/10.1038/ni974
- ↑ Kavli B, Andersen S, Otterlei M, Liabakk NB, Imai K, Fischer A, Durandy A, Krokan HE, Slupphaug G. B cells from hyper-IgM patients carrying UNG mutations lack ability to remove uracil from ssDNA and have elevated genomic uracil. J Exp Med. 2005 Jun 20;201(12):2011-21. PMID:15967827 doi:10.1084/jem.20050042
- ↑ Groisman R, Polanowska J, Kuraoka I, Sawada J, Saijo M, Drapkin R, Kisselev AF, Tanaka K, Nakatani Y. The ubiquitin ligase activity in the DDB2 and CSA complexes is differentially regulated by the COP9 signalosome in response to DNA damage. Cell. 2003 May 2;113(3):357-67. PMID:12732143
- ↑ Hu J, McCall CM, Ohta T, Xiong Y. Targeted ubiquitination of CDT1 by the DDB1-CUL4A-ROC1 ligase in response to DNA damage. Nat Cell Biol. 2004 Oct;6(10):1003-9. Epub 2004 Sep 26. PMID:15448697 doi:10.1038/ncb1172
- ↑ Wertz IE, O'Rourke KM, Zhang Z, Dornan D, Arnott D, Deshaies RJ, Dixit VM. Human De-etiolated-1 regulates c-Jun by assembling a CUL4A ubiquitin ligase. Science. 2004 Feb 27;303(5662):1371-4. Epub 2004 Jan 22. PMID:14739464 doi:10.1126/science.1093549
- ↑ Sugasawa K, Okuda Y, Saijo M, Nishi R, Matsuda N, Chu G, Mori T, Iwai S, Tanaka K, Tanaka K, Hanaoka F. UV-induced ubiquitylation of XPC protein mediated by UV-DDB-ubiquitin ligase complex. Cell. 2005 May 6;121(3):387-400. PMID:15882621 doi:10.1016/j.cell.2005.02.035
- ↑ Kulaksiz G, Reardon JT, Sancar A. Xeroderma pigmentosum complementation group E protein (XPE/DDB2): purification of various complexes of XPE and analyses of their damaged DNA binding and putative DNA repair properties. Mol Cell Biol. 2005 Nov;25(22):9784-92. PMID:16260596 doi:10.1128/MCB.25.22.9784-9792.2005
- ↑ Nishitani H, Sugimoto N, Roukos V, Nakanishi Y, Saijo M, Obuse C, Tsurimoto T, Nakayama KI, Nakayama K, Fujita M, Lygerou Z, Nishimoto T. Two E3 ubiquitin ligases, SCF-Skp2 and DDB1-Cul4, target human Cdt1 for proteolysis. EMBO J. 2006 Mar 8;25(5):1126-36. Epub 2006 Feb 16. PMID:16482215 doi:7601002
- ↑ He YJ, McCall CM, Hu J, Zeng Y, Xiong Y. DDB1 functions as a linker to recruit receptor WD40 proteins to CUL4-ROC1 ubiquitin ligases. Genes Dev. 2006 Nov 1;20(21):2949-54. PMID:17079684 doi:20/21/2949
- ↑ Hu J, Xiong Y. An evolutionarily conserved function of proliferating cell nuclear antigen for Cdt1 degradation by the Cul4-Ddb1 ubiquitin ligase in response to DNA damage. J Biol Chem. 2006 Feb 17;281(7):3753-6. Epub 2006 Jan 3. PMID:16407242 doi:10.1074/jbc.C500464200
- ↑ Senga T, Sivaprasad U, Zhu W, Park JH, Arias EE, Walter JC, Dutta A. PCNA is a cofactor for Cdt1 degradation by CUL4/DDB1-mediated N-terminal ubiquitination. J Biol Chem. 2006 Mar 10;281(10):6246-52. Epub 2006 Jan 9. PMID:16407252 doi:M512705200
- ↑ Wang H, Zhai L, Xu J, Joo HY, Jackson S, Erdjument-Bromage H, Tempst P, Xiong Y, Zhang Y. Histone H3 and H4 ubiquitylation by the CUL4-DDB-ROC1 ubiquitin ligase facilitates cellular response to DNA damage. Mol Cell. 2006 May 5;22(3):383-94. PMID:16678110 doi:S1097-2765(06)00230-9
- ↑ Lovejoy CA, Lock K, Yenamandra A, Cortez D. DDB1 maintains genome integrity through regulation of Cdt1. Mol Cell Biol. 2006 Nov;26(21):7977-90. Epub 2006 Aug 28. PMID:16940174 doi:MCB.00819-06
- ↑ Higa LA, Wu M, Ye T, Kobayashi R, Sun H, Zhang H. CUL4-DDB1 ubiquitin ligase interacts with multiple WD40-repeat proteins and regulates histone methylation. Nat Cell Biol. 2006 Nov;8(11):1277-83. Epub 2006 Oct 15. PMID:17041588 doi:10.1038/ncb1490
- ↑ Kapetanaki MG, Guerrero-Santoro J, Bisi DC, Hsieh CL, Rapic-Otrin V, Levine AS. The DDB1-CUL4ADDB2 ubiquitin ligase is deficient in xeroderma pigmentosum group E and targets histone H2A at UV-damaged DNA sites. Proc Natl Acad Sci U S A. 2006 Feb 21;103(8):2588-93. Epub 2006 Feb 10. PMID:16473935 doi:10.1073/pnas.0511160103
- ↑ Guerrero-Santoro J, Kapetanaki MG, Hsieh CL, Gorbachinsky I, Levine AS, Rapic-Otrin V. The cullin 4B-based UV-damaged DNA-binding protein ligase binds to UV-damaged chromatin and ubiquitinates histone H2A. Cancer Res. 2008 Jul 1;68(13):5014-22. PMID:18593899 doi:68/13/5014
- ↑ Hu J, Zacharek S, He YJ, Lee H, Shumway S, Duronio RJ, Xiong Y. WD40 protein FBW5 promotes ubiquitination of tumor suppressor TSC2 by DDB1-CUL4-ROC1 ligase. Genes Dev. 2008 Apr 1;22(7):866-71. doi: 10.1101/gad.1624008. PMID:18381890 doi:10.1101/gad.1624008
- ↑ Huang J, Chen J. VprBP targets Merlin to the Roc1-Cul4A-DDB1 E3 ligase complex for degradation. Oncogene. 2008 Jul 3;27(29):4056-64. Epub 2008 Mar 10. PMID:18332868 doi:onc200844
- ↑ Le Rouzic E, Belaidouni N, Estrabaud E, Morel M, Rain JC, Transy C, Margottin-Goguet F. HIV1 Vpr arrests the cell cycle by recruiting DCAF1/VprBP, a receptor of the Cul4-DDB1 ubiquitin ligase. Cell Cycle. 2007 Jan 15;6(2):182-8. Epub 2007 Jan 17. PMID:17314515
- ↑ Wen X, Duus KM, Friedrich TD, de Noronha CM. The HIV1 protein Vpr acts to promote G2 cell cycle arrest by engaging a DDB1 and Cullin4A-containing ubiquitin ligase complex using VprBP/DCAF1 as an adaptor. J Biol Chem. 2007 Sep 14;282(37):27046-57. Epub 2007 Jul 9. PMID:17620334 doi:http://dx.doi.org/10.1074/jbc.M703955200
- ↑ Tan L, Ehrlich E, Yu XF. DDB1 and Cul4A are required for human immunodeficiency virus type 1 Vpr-induced G2 arrest. J Virol. 2007 Oct;81(19):10822-30. Epub 2007 Jul 11. PMID:17626091 doi:http://dx.doi.org/10.1128/JVI.01380-07
- ↑ Belzile JP, Duisit G, Rougeau N, Mercier J, Finzi A, Cohen EA. HIV-1 Vpr-mediated G2 arrest involves the DDB1-CUL4AVPRBP E3 ubiquitin ligase. PLoS Pathog. 2007 Jul;3(7):e85. PMID:17630831 doi:http://dx.doi.org/10.1371/journal.ppat.0030085
- ↑ Hrecka K, Gierszewska M, Srivastava S, Kozaczkiewicz L, Swanson SK, Florens L, Washburn MP, Skowronski J. Lentiviral Vpr usurps Cul4-DDB1[VprBP] E3 ubiquitin ligase to modulate cell cycle. Proc Natl Acad Sci U S A. 2007 Jul 10;104(28):11778-83. Epub 2007 Jul 3. PMID:17609381 doi:http://dx.doi.org/10.1073/pnas.0702102104
- ↑ DeHart JL, Zimmerman ES, Ardon O, Monteiro-Filho CM, Arganaraz ER, Planelles V. HIV-1 Vpr activates the G2 checkpoint through manipulation of the ubiquitin proteasome system. Virol J. 2007 Jun 8;4:57. PMID:17559673 doi:http://dx.doi.org/10.1186/1743-422X-4-57
- ↑ Le Rouzic E, Morel M, Ayinde D, Belaidouni N, Letienne J, Transy C, Margottin-Goguet F. Assembly with the Cul4A-DDB1DCAF1 ubiquitin ligase protects HIV-1 Vpr from proteasomal degradation. J Biol Chem. 2008 Aug 1;283(31):21686-92. doi: 10.1074/jbc.M710298200. Epub 2008 , Jun 4. PMID:18524771 doi:http://dx.doi.org/10.1074/jbc.M710298200
- ↑ McCall CM, Miliani de Marval PL, Chastain PD 2nd, Jackson SC, He YJ, Kotake Y, Cook JG, Xiong Y. Human immunodeficiency virus type 1 Vpr-binding protein VprBP, a WD40 protein associated with the DDB1-CUL4 E3 ubiquitin ligase, is essential for DNA replication and embryonic development. Mol Cell Biol. 2008 Sep;28(18):5621-33. doi: 10.1128/MCB.00232-08. Epub 2008 Jul , 7. PMID:18606781 doi:http://dx.doi.org/10.1128/MCB.00232-08
- ↑ Huang J, Chen J. VprBP targets Merlin to the Roc1-Cul4A-DDB1 E3 ligase complex for degradation. Oncogene. 2008 Jul 3;27(29):4056-64. Epub 2008 Mar 10. PMID:18332868 doi:onc200844
- ↑ Srivastava S, Swanson SK, Manel N, Florens L, Washburn MP, Skowronski J. Lentiviral Vpx accessory factor targets VprBP/DCAF1 substrate adaptor for cullin 4 E3 ubiquitin ligase to enable macrophage infection. PLoS Pathog. 2008 May 9;4(5):e1000059. doi: 10.1371/journal.ppat.1000059. PMID:18464893 doi:http://dx.doi.org/10.1371/journal.ppat.1000059
- ↑ Bergamaschi A, Ayinde D, David A, Le Rouzic E, Morel M, Collin G, Descamps D, Damond F, Brun-Vezinet F, Nisole S, Margottin-Goguet F, Pancino G, Transy C. The human immunodeficiency virus type 2 Vpx protein usurps the CUL4A-DDB1 DCAF1 ubiquitin ligase to overcome a postentry block in macrophage infection. J Virol. 2009 May;83(10):4854-60. doi: 10.1128/JVI.00187-09. Epub 2009 Mar 4. PMID:19264781 doi:http://dx.doi.org/10.1128/JVI.00187-09
- ↑ Gramberg T, Sunseri N, Landau NR. Evidence for an activation domain at the amino terminus of simian immunodeficiency virus Vpx. J Virol. 2010 Feb;84(3):1387-96. doi: 10.1128/JVI.01437-09. Epub 2009 Nov 18. PMID:19923175 doi:http://dx.doi.org/10.1128/JVI.01437-09
- ↑ Lee JM, Lee JS, Kim H, Kim K, Park H, Kim JY, Lee SH, Kim IS, Kim J, Lee M, Chung CH, Seo SB, Yoon JB, Ko E, Noh DY, Kim KI, Kim KK, Baek SH. EZH2 generates a methyl degron that is recognized by the DCAF1/DDB1/CUL4 E3 ubiquitin ligase complex. Mol Cell. 2012 Nov 30;48(4):572-86. doi: 10.1016/j.molcel.2012.09.004. Epub 2012 , Oct 11. PMID:23063525 doi:http://dx.doi.org/10.1016/j.molcel.2012.09.004
- ↑ Wu Y, Zhou X, Barnes CO, DeLucia M, Cohen AE, Gronenborn AM, Ahn J, Calero G. The DDB1-DCAF1-Vpr-UNG2 crystal structure reveals how HIV-1 Vpr steers human UNG2 toward destruction. Nat Struct Mol Biol. 2016 Aug 29. doi: 10.1038/nsmb.3284. PMID:27571178 doi:http://dx.doi.org/10.1038/nsmb.3284
|