This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
6eny
From Proteopedia
(New page: ==Structure of the human PLC editing module== <StructureSection load='6eny' size='340' side='right' caption='6eny, resolution 5.80Å' scene=''> == Structural highl...) |
m (Protected "6eny" [edit=sysop:move=sysop]) |
Revision as of 06:16, 29 November 2017
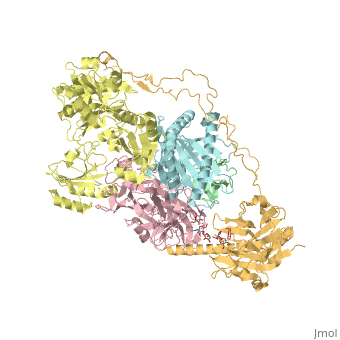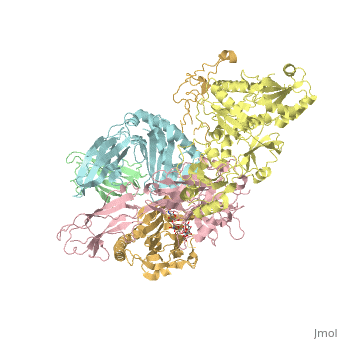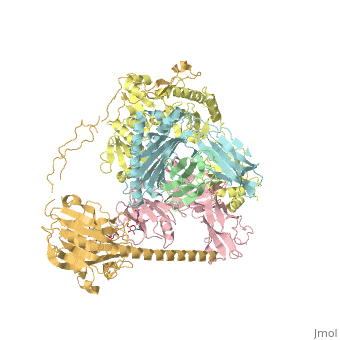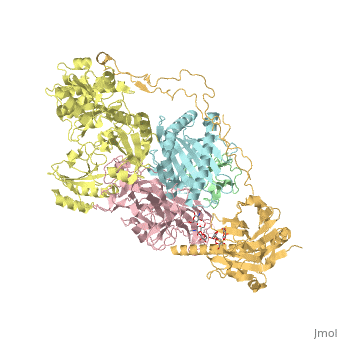
proteopedia linkproteopedia linkStructure of the human PLC editing module
Structural highlights
Disease[B2MG_HUMAN] Defects in B2M are the cause of hypercatabolic hypoproteinemia (HYCATHYP) [MIM:241600]. Affected individuals show marked reduction in serum concentrations of immunoglobulin and albumin, probably due to rapid degradation.[1] Note=Beta-2-microglobulin may adopt the fibrillar configuration of amyloid in certain pathologic states. The capacity to assemble into amyloid fibrils is concentration dependent. Persistently high beta(2)-microglobulin serum levels lead to amyloidosis in patients on long-term hemodialysis.[2] [3] [4] [5] [6] [7] [8] [9] [10] [11] [12] [13] [14] Function[1A03_HUMAN] Involved in the presentation of foreign antigens to the immune system. [B2MG_HUMAN] Component of the class I major histocompatibility complex (MHC). Involved in the presentation of peptide antigens to the immune system. [TPSN_HUMAN] Involved in the association of MHC class I with transporter associated with antigen processing (TAP) and in the assembly of MHC class I with peptide (peptide loading).[15] [CALR_HUMAN] Calcium-binding chaperone that promotes folding, oligomeric assembly and quality control in the endoplasmic reticulum (ER) via the calreticulin/calnexin cycle. This lectin interacts transiently with almost all of the monoglucosylated glycoproteins that are synthesized in the ER. Interacts with the DNA-binding domain of NR3C1 and mediates its nuclear export. Involved in maternal gene expression regulation. May participate in oocyte maturation via the regulation of calcium homeostasis (By similarity).[16] [17] Publication Abstract from PubMedThe peptide-loading complex (PLC) is a transient, multisubunit membrane complex in the endoplasmic reticulum that is essential for establishing a hierarchical immune response. The PLC coordinates peptide translocation into the endoplasmic reticulum with loading and editing of major histocompatibility complex class I (MHC-I) molecules. After final proofreading in the PLC, stable peptide-MHC-I complexes are released to the cell surface to evoke a T-cell response against infected or malignant cells. Sampling of different MHC-I allomorphs requires the precise coordination of seven different subunits in a single macromolecular assembly, including the transporter associated with antigen processing (TAP1 and TAP2, jointly referred to as TAP), the oxidoreductase ERp57, the MHC-I heterodimer, and the chaperones tapasin and calreticulin. The molecular organization of and mechanistic events that take place in the PLC are unknown owing to the heterogeneous composition and intrinsically dynamic nature of the complex. Here, we isolate human PLC from Burkitt's lymphoma cells using an engineered viral inhibitor as bait and determine the structure of native PLC by electron cryo-microscopy. Two endoplasmic reticulum-resident editing modules composed of tapasin, calreticulin, ERp57, and MHC-I are centred around TAP in a pseudo-symmetric orientation. A multivalent chaperone network within and across the editing modules establishes the proofreading function at two lateral binding platforms for MHC-I molecules. The lectin-like domain of calreticulin senses the MHC-I glycan, whereas the P domain reaches over the MHC-I peptide-binding pocket towards ERp57. This arrangement allows tapasin to facilitate peptide editing by clamping MHC-I. The translocation pathway of TAP opens out into a large endoplasmic reticulum lumenal cavity, confined by the membrane entry points of tapasin and MHC-I. Two lateral windows channel the antigenic peptides to MHC-I. Structures of PLC captured at distinct assembly states provide mechanistic insight into the recruitment and release of MHC-I. Our work defines the molecular symbiosis of an ABC transporter and an endoplasmic reticulum chaperone network in MHC-I assembly and provides insight into the onset of the adaptive immune response. Structure of the human MHC-I peptide-loading complex.,Blees A, Januliene D, Hofmann T, Koller N, Schmidt C, Trowitzsch S, Moeller A, Tampe R Nature. 2017 Nov 23;551(7681):525-528. doi: 10.1038/nature24627. Epub 2017 Nov 6. PMID:29107940[18] From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine. References
| ||||||||||||||||||||