Platelet-derived growth factors and receptors
From Proteopedia
(Difference between revisions)
| Line 16: | Line 16: | ||
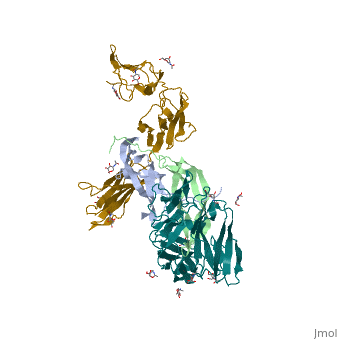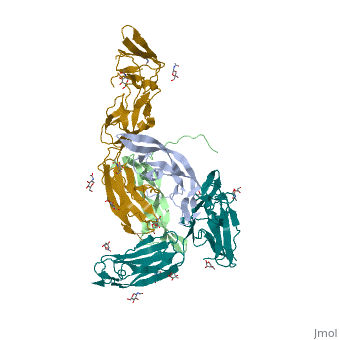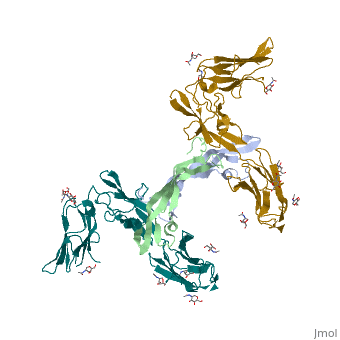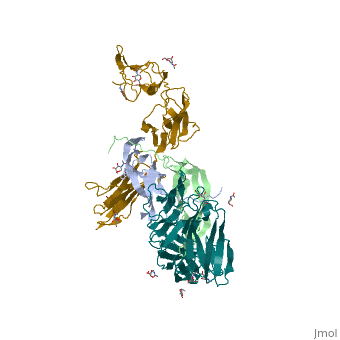
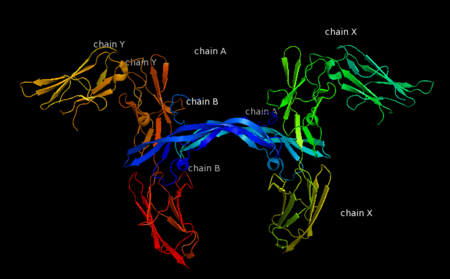
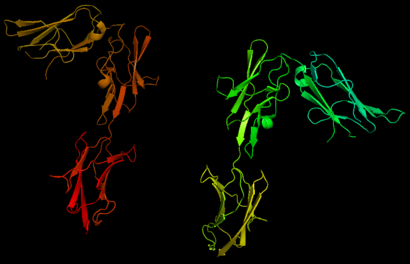
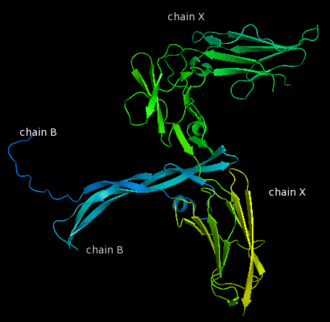
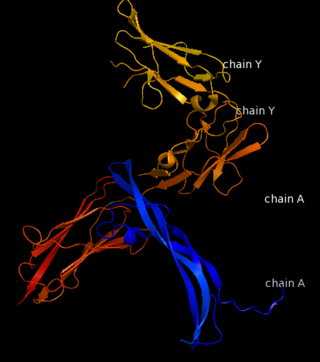
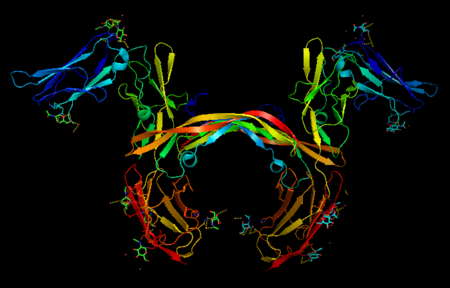
The PDGF family consists of PDGF-A, -B, -C and -D, which form either homo- or heterodimers (PDGF-AA, -AB, -BB, -CC, -DD). These four are inactive in their monomeric forms. However the PDGFs bind to the protein tyrosine kinase receptors PDGF receptor-α and -β. These two receptor isoforms dimerize upon binding of the PDGF dimer, leading to three possible receptor combinations, namely -αα, -ββ and -αβ. The extracellular region of the receptor consists of five immunoglobulin-type domains while the intracellular part is a tyrosine kinase domain. The ligand-binding sites of the receptors are located to the three first immunoglobulin type domains. PDGF-CC specifically interacts with PDGFR-αα and -αβ, but not with -ββ, and thereby resembles PDGF-AB. PDGF-DD binds to PDGFR-ββ with high affinity, and to PDGFR-αβ to a markedly lower extent and is therefore regarded as PDGFR-ββ specific. PDGF-AA binds only to PDGFR-αα, while PDGF-BB is the only PDGF that can bind all three receptor combinations with high affinity. | The PDGF family consists of PDGF-A, -B, -C and -D, which form either homo- or heterodimers (PDGF-AA, -AB, -BB, -CC, -DD). These four are inactive in their monomeric forms. However the PDGFs bind to the protein tyrosine kinase receptors PDGF receptor-α and -β. These two receptor isoforms dimerize upon binding of the PDGF dimer, leading to three possible receptor combinations, namely -αα, -ββ and -αβ. The extracellular region of the receptor consists of five immunoglobulin-type domains while the intracellular part is a tyrosine kinase domain. The ligand-binding sites of the receptors are located to the three first immunoglobulin type domains. PDGF-CC specifically interacts with PDGFR-αα and -αβ, but not with -ββ, and thereby resembles PDGF-AB. PDGF-DD binds to PDGFR-ββ with high affinity, and to PDGFR-αβ to a markedly lower extent and is therefore regarded as PDGFR-ββ specific. PDGF-AA binds only to PDGFR-αα, while PDGF-BB is the only PDGF that can bind all three receptor combinations with high affinity. | ||
| - | Dimerization is a prerequisite for the activation of the kinase. Kinase activation is visualized as tyrosine phosphorylation of the receptor molecules, which occurs between the dimerized receptor molecules (transphosphorylation). In conjunction with dimerization and kinase activation, the receptor molecules undergo conformational changes, which allow a basal kinase activity to phosphorylate a critical tyrosine residue, thereby "unlocking" the kinase, leading to full enzymatic activity directed toward other tyrosine residues in the receptor molecules as well as other substrates for the kinase. This ultimately effects gene expression and cell growth. Expression of both receptors and each of the four PDGFs is under independent control, giving the PDGF/PDGFR system a high flexibility. Different cell types vary greatly in the ratio of PDGF isoforms and PDGFRs expressed. Different external stimuli such as inflammation, embryonic development or differentiation modulate cellular receptor expression allowing binding of some PDGFs but not others. Additionally, some cells display only one of the PDGFR isoforms while other cells express both isoforms, simultaneously or separately. | + | Dimerization is a prerequisite for the activation of the kinase. Kinase activation is visualized as tyrosine phosphorylation of the receptor molecules, which occurs between the dimerized receptor molecules (transphosphorylation). In conjunction with dimerization and kinase activation, the receptor molecules undergo conformational changes, which allow a basal kinase activity to phosphorylate a critical tyrosine residue, thereby "unlocking" the kinase, leading to full enzymatic activity directed toward other tyrosine residues in the receptor molecules as well as other substrates for the kinase. This ultimately effects gene expression and cell growth. Expression of both receptors and each of the four PDGFs is under independent control, giving the PDGF/PDGFR system a high flexibility. Different cell types vary greatly in the ratio of PDGF isoforms and PDGFRs expressed. Different external stimuli such as inflammation, embryonic development or differentiation modulate cellular receptor expression allowing binding of some PDGFs but not others. Additionally, some cells display only one of the PDGFR isoforms while other cells express both isoforms, simultaneously or separately<ref>PMID:2543106</ref> . |
Tyrosine phosphorylation sites in growth factor receptors serve two major purposes: to control the state of activity of the kinase and to create binding sites for downstream signal transduction molecules, which in many cases also are substrates for the kinase. The second part of the tyrosine kinase domain in the PDGFβ receptor is phosphorylated at Tyr-857, and mutant receptors carrying phenylalanine at this position have reduced kinase activity. Tyr-857 has therefore been assigned a role in positive regulation of kinase activity. | Tyrosine phosphorylation sites in growth factor receptors serve two major purposes: to control the state of activity of the kinase and to create binding sites for downstream signal transduction molecules, which in many cases also are substrates for the kinase. The second part of the tyrosine kinase domain in the PDGFβ receptor is phosphorylated at Tyr-857, and mutant receptors carrying phenylalanine at this position have reduced kinase activity. Tyr-857 has therefore been assigned a role in positive regulation of kinase activity. | ||
Revision as of 21:57, 19 January 2018
| |||||||||||
References
- ↑ Heldin CH, Ostman A, Eriksson A, Siegbahn A, Claesson-Welsh L, Westermark B. Platelet-derived growth factor: isoform-specific signalling via heterodimeric or homodimeric receptor complexes. Kidney Int. 1992 Mar;41(3):571-4. PMID:1315403
- ↑ Shim AH, Liu H, Focia PJ, Chen X, Lin PC, He X. Structures of a platelet-derived growth factor/propeptide complex and a platelet-derived growth factor/receptor complex. Proc Natl Acad Sci U S A. 2010 Jun 22;107(25):11307-12. Epub 2010 Jun 2. PMID:20534510
- ↑ Heldin CH, Westermark B. Platelet-derived growth factor: three isoforms and two receptor types. Trends Genet. 1989 Apr;5(4):108-11. PMID:2543106
- ↑ Williams LT. Signal transduction by the platelet-derived growth factor receptor. Science. 1989 Mar 24;243(4898):1564-70. PMID:2538922
<5>Heldin CH, Westermark B (April 1989). "Platelet-derived growth factor: three isoforms and two receptor types". Trends Genet. 5 (4): 108–11. DOI:10.1016/0168-9525(89)90040-1. PMID 2543106.
Proteopedia Page Contributors and Editors (what is this?)
Joel L. Sussman, Lisa Tice, Michal Harel, Angel Herraez, Jaime Prilusky, Alexander Berchansky