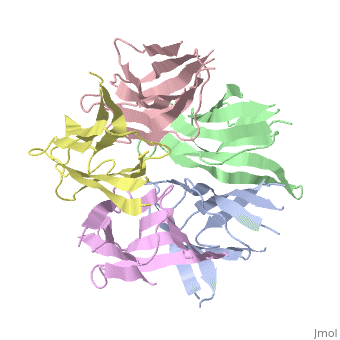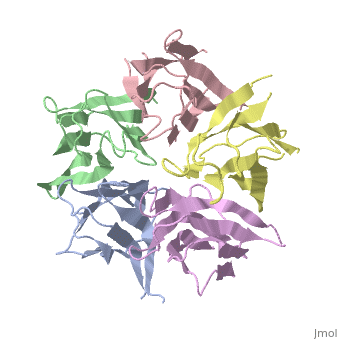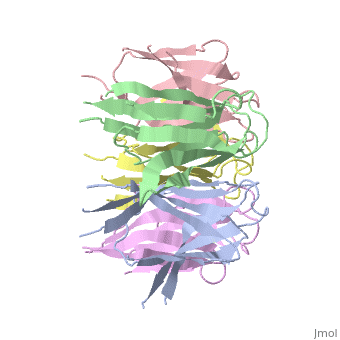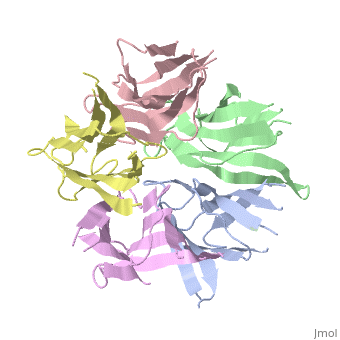This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Nucleoplasmin
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
=Structural highlights= | =Structural highlights= | ||
Nucleoplasmin (NP) is made out of five <scene name='46/467273/Np_monomer/1'>monomers</scene>,that create ring-shaped histone chaperone. The monomers are formed by a <scene name='46/467273/Core_domain/1'>core domain</scene> that responsible for oligomerization, that make the protein highly stable and compact. | Nucleoplasmin (NP) is made out of five <scene name='46/467273/Np_monomer/1'>monomers</scene>,that create ring-shaped histone chaperone. The monomers are formed by a <scene name='46/467273/Core_domain/1'>core domain</scene> that responsible for oligomerization, that make the protein highly stable and compact. | ||
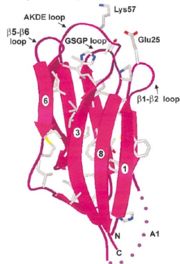
| - | The NP core is made out of eight | + | The NP core is made out of eight β strands that form a barrel with a jellyroll topology. Residues in the hydrophobic core are highly conserved (aproximate 80%). The AKDE and GSGP motifs are ordered loops and may function in decamer formation. |
[[Image:NP core.jpg|thumb|NP core]] | [[Image:NP core.jpg|thumb|NP core]] | ||
| Line 56: | Line 56: | ||
the NP in Humans is made out of <scene name='46/467273/Dimer/1'>dimer</scene> while each <scene name='46/467273/Monomer/2'>monomer</scene> consisted of five chains. The structure remain similiar to the one in Xenopus but with a change in amino acids in its <scene name='46/467273/Core_domain/2'>core domain</scene>, Val insted of Ile. | the NP in Humans is made out of <scene name='46/467273/Dimer/1'>dimer</scene> while each <scene name='46/467273/Monomer/2'>monomer</scene> consisted of five chains. The structure remain similiar to the one in Xenopus but with a change in amino acids in its <scene name='46/467273/Core_domain/2'>core domain</scene>, Val insted of Ile. | ||
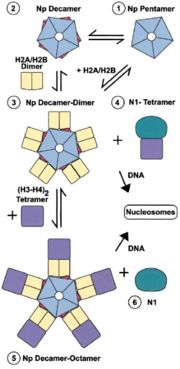
The Decamer bind H2A-H2B dimers and H3-H4 tetramers simultaneously, In the absence of histone tetramers the pentamer binds H2A-H2B and formes central hub. When H3-H4 tetramers are recruited this results in a functional dimerization of the complex, and the decamer being formed. | The Decamer bind H2A-H2B dimers and H3-H4 tetramers simultaneously, In the absence of histone tetramers the pentamer binds H2A-H2B and formes central hub. When H3-H4 tetramers are recruited this results in a functional dimerization of the complex, and the decamer being formed. | ||
| + | [[Image:Histone Storage and Nucleosome Assembly.jpg|thumb|Histone Storage and Nucleosome Assembly]] | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
== conservastion == | == conservastion == | ||
| Line 106: | Line 139: | ||
6. Sonia Ban˜ uelos,Miren J. Omaetxebarria, Isbaal Ramos,Martin R. Larsen, Igor Arregi, Ole N. Jensen, Jesus M. Arizmendi, Adelina Prado, and Arturo Muga. Phosphorylation of Both Nucleoplasmin Domains Is Required | 6. Sonia Ban˜ uelos,Miren J. Omaetxebarria, Isbaal Ramos,Martin R. Larsen, Igor Arregi, Ole N. Jensen, Jesus M. Arizmendi, Adelina Prado, and Arturo Muga. Phosphorylation of Both Nucleoplasmin Domains Is Required | ||
for Activation of Its Chromatin Decondensation Activity. THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 282, NO. 29, pp. 21213–21221, July 20, 2007 | for Activation of Its Chromatin Decondensation Activity. THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 282, NO. 29, pp. 21213–21221, July 20, 2007 | ||
| + | |||
| + | 7. Shuchismita Dutta, Ildiko´ V. Akey,Colin Dingwall, Kari L. Hartman,Tom Laue, Robert T. Nolte,James F. Head, and Christopher W. Akey. The Crystal Structure of Nucleoplasmin-Core: | ||
| + | Implications for Histone Binding and Nucleosome Assembly. Molecular Cell, Vol. 8, 841–853, October, 2001 | ||
Revision as of 16:51, 11 March 2018
| |||||||||||