User:David Ryskamp/Sandbox1
From Proteopedia
(Difference between revisions)
| Line 16: | Line 16: | ||
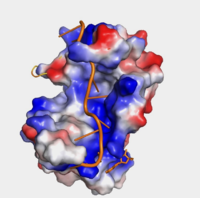
The <scene name='78/782616/Subunits_of_pabp/3'>Components of PABP</scene> are categorized into two RRMs: the n-terminus RRM1 (red) and c-terminus RRM2 (blue) are shown accordingly. The two RRMs are linked via an alpha-helix linker (green) that maintains the RRM1/2 complex that is the biological assembly and active form of PABP. Each RRM has a four-stranded antiparallel beta sheet backed by two corresponding alpha helices. <ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> mRNA poly-adenosine recognition is due to the presence of the conserved residues within the beta-sheet surface <ref name="The Poly(A)-Binding Protein and an mRNA Stability Protein Jointly Regulate an Endoribonuclease Activity.">Wang, Zuoren and Kiledjian, Megerditch. “The Poly(A)-Binding Protein and an mRNA Stability Protein Jointly Regulate an Endoribonuclease Activity.” Molecular and Cellular Biology 20.17 (2000): 6334–6341. Print.</ref> , which forms a <scene name='78/782616/Trough2/1'>trough</scene>-like pocket for the mRNA to bind. The beta-sheet flooring present in PABP interacts with the 3’ mRNA tail via a combination of van der Waals, aromatic stacking, and Hydrogen bonding. Three major aromatic stacking interactions are <scene name='78/782616/A3-phe102/1'>Phe102 with A3</scene>, <scene name='78/782616/Tyr56-a8/1'>Tyr56 with A8</scene>, and <scene name='78/782616/A6-tyr14/1'>Tyr14 with A6</scene>. Through these interactions, PABP binds to 3’ Poly (A) tail with a KD of 2-7 nM. <ref name="Roles of Cytoplasmic Poly(A)-Binding Proteins">Gorgoni, Barbra, and Gray, Nicola. “The Roles of Cytoplasmic Poly(A)-Binding Proteins in Regulating Gene Expression: A Developmental Perspective.” Briefings in Functional Genomics and Proteomics, vol. 3, no. 2, 1 Aug. 2004, pp. 125–141., doi:10.1093/bfgp/3.2.125.</ref> [[Image:Hydrophobicity (1).png|200px|right|thumb| "Figure 1:" Surface hydrophobicity shown in presence of mRNA]] | The <scene name='78/782616/Subunits_of_pabp/3'>Components of PABP</scene> are categorized into two RRMs: the n-terminus RRM1 (red) and c-terminus RRM2 (blue) are shown accordingly. The two RRMs are linked via an alpha-helix linker (green) that maintains the RRM1/2 complex that is the biological assembly and active form of PABP. Each RRM has a four-stranded antiparallel beta sheet backed by two corresponding alpha helices. <ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> mRNA poly-adenosine recognition is due to the presence of the conserved residues within the beta-sheet surface <ref name="The Poly(A)-Binding Protein and an mRNA Stability Protein Jointly Regulate an Endoribonuclease Activity.">Wang, Zuoren and Kiledjian, Megerditch. “The Poly(A)-Binding Protein and an mRNA Stability Protein Jointly Regulate an Endoribonuclease Activity.” Molecular and Cellular Biology 20.17 (2000): 6334–6341. Print.</ref> , which forms a <scene name='78/782616/Trough2/1'>trough</scene>-like pocket for the mRNA to bind. The beta-sheet flooring present in PABP interacts with the 3’ mRNA tail via a combination of van der Waals, aromatic stacking, and Hydrogen bonding. Three major aromatic stacking interactions are <scene name='78/782616/A3-phe102/1'>Phe102 with A3</scene>, <scene name='78/782616/Tyr56-a8/1'>Tyr56 with A8</scene>, and <scene name='78/782616/A6-tyr14/1'>Tyr14 with A6</scene>. Through these interactions, PABP binds to 3’ Poly (A) tail with a KD of 2-7 nM. <ref name="Roles of Cytoplasmic Poly(A)-Binding Proteins">Gorgoni, Barbra, and Gray, Nicola. “The Roles of Cytoplasmic Poly(A)-Binding Proteins in Regulating Gene Expression: A Developmental Perspective.” Briefings in Functional Genomics and Proteomics, vol. 3, no. 2, 1 Aug. 2004, pp. 125–141., doi:10.1093/bfgp/3.2.125.</ref> [[Image:Hydrophobicity (1).png|200px|right|thumb| "Figure 1:" Surface hydrophobicity shown in presence of mRNA]] | ||
| - | Further, the RRM1/2 complex interacts with the mRNA's sugar-phosphate backbone, where 4 of the 8 mRNA adenosines interact electrostatically.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> Upon closer examination of the PABP structure, the protein contains loop-like domains that form the walls of the beta-sheet trough. Although these <scene name='78/782616/Walls_of_trough/3'>loop walls</scene> are present, no interaction occurs between the mRNA and these regions. We propose that these loops only keep unwanted cellular elements out of the binding pocket via hydrophobic and hydrophilic interactions, maintaining the protein's selectivity for mRNA (Figure 1). The structural elements highlighted consist of the RRM1/2 subunits, the linker domain, and the Poly(A) mRNA binding trough. | ||
| + | |||
| + | Further, the RRM1/2 complex interacts with the mRNA's sugar-phosphate backbone, where 4 of the 8 mRNA adenosines interact electrostatically.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> Upon closer examination of the PABP structure, the protein contains loop-like domains that form the walls of the beta-sheet trough. Although these <scene name='78/782616/Walls_of_trough/3'>loop walls</scene> are present, no interaction occurs between the mRNA and these regions. We propose that these loops only keep unwanted cellular elements out of the binding pocket via hydrophobic and hydrophilic interactions, maintaining the protein's selectivity for mRNA (Figure 1). The structural elements highlighted consist of the RRM1/2 subunits, the linker domain, and the Poly(A) mRNA binding trough. | ||
===Interactions=== | ===Interactions=== | ||
Revision as of 17:37, 6 April 2018
Human Poly(A) Binding Protein (1CVJ)
| |||||||||||
References
- ↑ Blobel, Gunter. “A Protein of Molecular Weight 78,000 Bound to the Polyadenylate Region of Eukaryotic Messenger Rnas.” Proceedings of the National Academy of Sciences of the United States of America, vol. 70, no. 3, 1973, pp. 924–8.
- ↑ Baer, Bradford W. and Kornberg, Roger D. "The Protein Responsible for the Repeating Structure of Cytoplasmic Poly(A)-Ribonucleoprotein." The Journal of Cell Biology, vol. 96, no. 3, Mar. 1983, pp. 717-721. EBSCOhost.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 3.16 Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print.
- ↑ Kühn, Uwe and Elmar, Wahle. “Structure and Function of Poly(a) Binding Proteins.” Bba - Gene Structure & Expression, vol. 1678, no. 2/3, 2004.
- ↑ 5.0 5.1 5.2 5.3 5.4 Gorgoni, Barbra, and Gray, Nicola. “The Roles of Cytoplasmic Poly(A)-Binding Proteins in Regulating Gene Expression: A Developmental Perspective.” Briefings in Functional Genomics and Proteomics, vol. 3, no. 2, 1 Aug. 2004, pp. 125–141., doi:10.1093/bfgp/3.2.125.
- ↑ Wang, Zuoren and Kiledjian, Megerditch. “The Poly(A)-Binding Protein and an mRNA Stability Protein Jointly Regulate an Endoribonuclease Activity.” Molecular and Cellular Biology 20.17 (2000): 6334–6341. Print.
- ↑ 7.0 7.1 7.2 7.3 “Oculopharyngeal Muscular Dystrophy.” NORD (National Organization for Rare Disorders), rarediseases.org/rare-diseases/oculopharyngeal-muscular-dystrophy/.
- ↑ Richard, Pascale, et al. “Correlation between PABPN1 Genotype and Disease Severity in Oculopharyngeal Muscular Dystrophy.” Neurology, vol. 88, no. 4, 2016, pp. 359–365., doi:10.1212/wnl.0000000000003554.