This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Ethan Kitt/Sandbox 1
From Proteopedia
| Line 8: | Line 8: | ||
<StructureSection load='1B7F' size='350' frame='true' side='right' caption='PABP' scene='78/782614/Structure_scene_color_scheme/1'> | <StructureSection load='1B7F' size='350' frame='true' side='right' caption='PABP' scene='78/782614/Structure_scene_color_scheme/1'> | ||
The crystal structure PABP was derived from X-ray Diffraction at 2.6Å (R-value: 23%). The subunits of PABP, RRM1 and RRM2, are examined in this article as the ''in vivo'' form seen in biological assembly 1 (via PDB). The protein has a homopolymeric structure, containing four RNA recognition motifs (RRMs), which are conserved. <ref name="Structure and Function">Kühn, Uwe and Elmar, Wahle. “Structure and Function of Poly(a) Binding Proteins.” Bba - Gene Structure & Expression, vol. 1678, no. 2/3, 2004. </ref> <scene name='78/782616/Rrm1_only/2'>RRM1</scene> and <scene name='78/782616/Rrm2_only/2'>RRM2</scene> are N-terminal domains that are connected by a <scene name='78/782616/Linker/3'>linker</scene>.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> Opposed to their counterparts, RRM3 and RRM4 bind Poly (A) RNA less tightly than RRM1 and RRM2.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> | The crystal structure PABP was derived from X-ray Diffraction at 2.6Å (R-value: 23%). The subunits of PABP, RRM1 and RRM2, are examined in this article as the ''in vivo'' form seen in biological assembly 1 (via PDB). The protein has a homopolymeric structure, containing four RNA recognition motifs (RRMs), which are conserved. <ref name="Structure and Function">Kühn, Uwe and Elmar, Wahle. “Structure and Function of Poly(a) Binding Proteins.” Bba - Gene Structure & Expression, vol. 1678, no. 2/3, 2004. </ref> <scene name='78/782616/Rrm1_only/2'>RRM1</scene> and <scene name='78/782616/Rrm2_only/2'>RRM2</scene> are N-terminal domains that are connected by a <scene name='78/782616/Linker/3'>linker</scene>.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> Opposed to their counterparts, RRM3 and RRM4 bind Poly (A) RNA less tightly than RRM1 and RRM2.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> | ||
| - | ===RRMs=== | + | ===RNA Recognition Motifs (RRMs)=== |
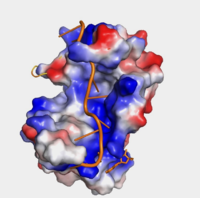
| - | + | The <scene name='78/782616/Subunits_of_pabp/3'>Components of PABP</scene> are categorized into two RRMs: the n-terminus RRM1 (red) and c-terminus RRM2 (blue) are shown accordingly. The two RRMs are linked via an alpha-helix linker (green) that maintains the RRM1/2 complex that is the biological assembly and active form of PABP. Each RRM has a four-stranded antiparallel beta sheet backed by two corresponding alpha helices. <ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> mRNA poly-adenosine recognition is due to the presence of the conserved residues within the beta-sheet surface <ref name="The Poly(A)-Binding Protein and an mRNA Stability Protein Jointly Regulate an Endoribonuclease Activity.">Wang, Zuoren and Kiledjian, Megerditch. “The Poly(A)-Binding Protein and an mRNA Stability Protein Jointly Regulate an Endoribonuclease Activity.” Molecular and Cellular Biology 20.17 (2000): 6334–6341. Print.</ref> , which forms a <scene name='78/782616/Trough2/1'>trough</scene>-like pocket for the mRNA to bind. The beta-sheet flooring present in PABP interacts with the 3’ mRNA tail via a combination of van der Waals, aromatic stacking, and Hydrogen bonding. Through these interactions, PABP binds to 3’ Poly (A) tail with a KD of 2-7 nM. <ref name="Roles of Cytoplasmic Poly(A)-Binding Proteins">Gorgoni, Barbra, and Gray, Nicola. “The Roles of Cytoplasmic Poly(A)-Binding Proteins in Regulating Gene Expression: A Developmental Perspective.” Briefings in Functional Genomics and Proteomics, vol. 3, no. 2, 1 Aug. 2004, pp. 125–141., doi:10.1093/bfgp/3.2.125.</ref> [[Image:Hydrophobicity (1).png|200px|right|thumb| "Figure 1:" Surface hydrophobicity shown in presence of mRNA]] | |
=== The Ligand=== | === The Ligand=== | ||
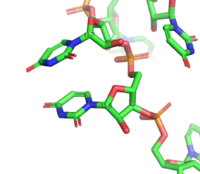
[[Image:Stacking.png|200 px|right|thumb|Figure 3: Intramolecular ring stacking of U7 (top) and U8 (bottom)]] The RNA <scene name='78/782599/Ligand_only/4'>ligand</scene> bound by Sxl is 9 nucleotides long—UGUUUUUUU. This ligand lacks intramolecular base pairs—a characteristic that would typically assist in RNA recognition—and therefore presents with many unique features. Sxl fixes U3-U11 in a specific elongated conformation. <scene name='78/782599/U6-11/4'>U6-U11</scene> interact within the strongly electropositive v-shaped cleft, while <scene name='78/782600/U3-u5/7'>U3-G4-U5</scene> are bound to a positively charged surface on RRM2 <ref name="Handa" />. The kink in the middle of the ligand is created by hydrogen bonds between the 2’OH's of U5 and U6 and the phosphate group of U8, as well as between the 2’OH of U7 and the phosphate group of U5. This is kink is also facilitated by the only intramolecular stacking pair in the ligand, U7 and U8 (Figure 3). All of the nucleotides besides U8 are in the [http://x3dna.org/highlights/sugar-pucker-correlates-with-phosphorus-base-distance C2’-endo] conformation. This orientation allows the bases to be highly exposed to the protein and therefore increases specificity. The low number of intramolecular stacking regions and the large number of C2’-endo conformations deem this ligand unique<ref name="Handa" />. | [[Image:Stacking.png|200 px|right|thumb|Figure 3: Intramolecular ring stacking of U7 (top) and U8 (bottom)]] The RNA <scene name='78/782599/Ligand_only/4'>ligand</scene> bound by Sxl is 9 nucleotides long—UGUUUUUUU. This ligand lacks intramolecular base pairs—a characteristic that would typically assist in RNA recognition—and therefore presents with many unique features. Sxl fixes U3-U11 in a specific elongated conformation. <scene name='78/782599/U6-11/4'>U6-U11</scene> interact within the strongly electropositive v-shaped cleft, while <scene name='78/782600/U3-u5/7'>U3-G4-U5</scene> are bound to a positively charged surface on RRM2 <ref name="Handa" />. The kink in the middle of the ligand is created by hydrogen bonds between the 2’OH's of U5 and U6 and the phosphate group of U8, as well as between the 2’OH of U7 and the phosphate group of U5. This is kink is also facilitated by the only intramolecular stacking pair in the ligand, U7 and U8 (Figure 3). All of the nucleotides besides U8 are in the [http://x3dna.org/highlights/sugar-pucker-correlates-with-phosphorus-base-distance C2’-endo] conformation. This orientation allows the bases to be highly exposed to the protein and therefore increases specificity. The low number of intramolecular stacking regions and the large number of C2’-endo conformations deem this ligand unique<ref name="Handa" />. | ||
Revision as of 18:11, 20 April 2018
Contents |
Human Poly(A) Binding Protein (1CVJ)
Background
The Human Poly(A) Binding Protein (PABP) was discovered in 1973 by the use of a sedimentation profile detailing the RNase digestion differentiated the PABP protein. [1] Attempts to purify the 75 kDa protein then followed. In 1983, then considered “poly(A)-organizing protein,” was determined and purified by molecular weight, ligand-binding affinity, and amounts found in cytoplasmic portions of cell with ability to bind to free poly(A). [2]
Structure
| |||||||||||
Autoregulation
Sxl controls its own levels of expression via positive and negative autoregulation. Sxl binds its own pre-mRNA transcript in a similar manner as its downstream targets, Tra and Msl2. Through binding to its recognition element, Sxl causes a 3’ splice site to be skipped. Alternative splicing occurs utilizing a 3’ splice site further downstream, cleaving out a premature stop codon within Exon 3 and preventing truncation and inactivation[8]. This is a pathway of positive autoregulation, as functional Sxl protein must be present to cause proper processing of the pre-mRNA. The negative autoregulation pathway of Sxl proceeds via repression of its own translation. The Sxl transcript contains the target polyuridine sequence within its 3’UTR. Sxl binds this target, and blocks translation[8]. Negative autoregulation allows maintenance of a stable and standard Sxl protein concentration. An excess of Sxl increases the degree of translation repression because more Sxl protein are present to potentially bind at the 3’UTR, while a shortage allows for more unrepressed translation.
Mutation
In fruit flies, dosage compensation is achieved by hyperexpression of the single X chromosome found in the male genome. The single X chromosome is hyperexpressed and regulated by a group of genes known as the Male-specific lethal genes. The genes involve work together to produce a complex known as the Male-specific lethal complex (Msl complex). One gene that is important in forming this complex is the gene that codes for the Msl-2 protein. Without this protein, the Msl complex cannot form [9]. Males exclusively make the Msl-2 protein and therefore exclusively create the Msl complex. Female cannot make the complex because the Sxl protein prevents the production of the Msl-2 protein. It prevents the protein formation by alternatively splicing of an intron near the 5’ UTR. In males this intron is included and in females it is not. [9] Mutations of the Sxl protein can affect the formation of this complex and lead to death for females—hence the name sex lethal. If the Sxl gene is mutated in females, the formation of the Msl-2 complex will not be inhibited. If there is a functional Msl-2 complex in females then both of the X chromosomes will be hyperexpressed [9] The overexpression of genes on the X chromosome is a fatal phenomenon ultimately caused by a mutation of the Sxl protein. Although a mutation to the Sxl gene is harmful to females, it has no effect on males because the functional Sxl gene is not expressed—a mutation in males has no phenotypic effect.
References
- ↑ Blobel, Gunter. “A Protein of Molecular Weight 78,000 Bound to the Polyadenylate Region of Eukaryotic Messenger Rnas.” Proceedings of the National Academy of Sciences of the United States of America, vol. 70, no. 3, 1973, pp. 924–8.
- ↑ Baer, Bradford W. and Kornberg, Roger D. "The Protein Responsible for the Repeating Structure of Cytoplasmic Poly(A)-Ribonucleoprotein." The Journal of Cell Biology, vol. 96, no. 3, Mar. 1983, pp. 717-721. EBSCOhost.
- ↑ Kühn, Uwe and Elmar, Wahle. “Structure and Function of Poly(a) Binding Proteins.” Bba - Gene Structure & Expression, vol. 1678, no. 2/3, 2004.
- ↑ 4.0 4.1 4.2 Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print.
- ↑ Wang, Zuoren and Kiledjian, Megerditch. “The Poly(A)-Binding Protein and an mRNA Stability Protein Jointly Regulate an Endoribonuclease Activity.” Molecular and Cellular Biology 20.17 (2000): 6334–6341. Print.
- ↑ Gorgoni, Barbra, and Gray, Nicola. “The Roles of Cytoplasmic Poly(A)-Binding Proteins in Regulating Gene Expression: A Developmental Perspective.” Briefings in Functional Genomics and Proteomics, vol. 3, no. 2, 1 Aug. 2004, pp. 125–141., doi:10.1093/bfgp/3.2.125.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedHanda - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedBlack - ↑ 9.0 9.1 9.2 Penalva L, Sanchez L. RNA Binding Protein Sex-Lethal (Sxl) and Control of Drosophila Sex Determination and Dosage Compensation. Microbiol Mol Biol Rev.;67(3):343-356. doi: 10.1128/MMBR.67.3.343–359.2003