This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:David Ryskamp/Sandbox1
From Proteopedia
| Line 56: | Line 56: | ||
===mRNA Stabilization=== | ===mRNA Stabilization=== | ||
| - | Serving yet again as another | + | Serving yet again as another source of mRNA stabilization, PABP prevents the deadenylation and decapping of the target mRNA. Typically, Poly(A) ribonuclease [https://en.wikipedia.org/wiki/Poly(A)-specific_ribonuclease (PARN)] works to deadenylate mRNA, but the presence of PABP hinders its function. The PABP protein is able to protect mRNA degradation through the complex that it forms with the elongation initiation factors, whose presence prevent deadenylation and decapping.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> This has been verified by the presence of deadenylation products and the comparative size of PABP footprints. There is some evidence indicating that PABP is involved in the prevention of endonucleolytic cleavage; however, only a small amount of mRNA is degraded from endonucleolytic cleavage, so it has not been widely researched.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> |
===Eukaryotic Translation Initiation=== | ===Eukaryotic Translation Initiation=== | ||
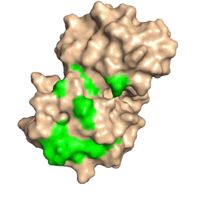
[[Image:Dorsal side.jpg|200px|right|thumb| "Figure 2:"Dorsal side with green conserved residues that interact with eIF4.]] | [[Image:Dorsal side.jpg|200px|right|thumb| "Figure 2:"Dorsal side with green conserved residues that interact with eIF4.]] | ||
| - | Upon mRNA Poly(A) recognition, PABP and the bound mRNA stimulate the initiation of translation by interacting with initiation factor eIF4G. Protein eIF4G | + | Upon mRNA Poly(A) recognition, PABP and the bound mRNA stimulate the initiation of translation by interacting with initiation factor eIF4G. Protein eIF4G interacts with PABP's dorsal side, under the trough, where hydrophobic and acidic residues stimulate the interaction between the two proteins (Figure 2). These specific residues are phylogenetically conserved among all PABPs, and are significant in the protein's function and interaction with eIF4G. |
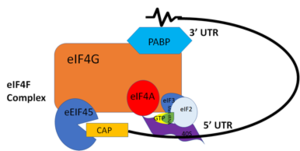
| - | PABP and mRNA complex aids in translation initiation under two proposed mechanisms. | + | PABP and mRNA complex aids in translation initiation under two proposed mechanisms. Studies have highlighted the presence of a “Closed Loop” Model in line with either mechanism. The “Closed Loop” Model entails the recognition of the 5’ 7-methyl-Guanosine cap by [https://en.wikipedia.org/wiki/Eukaryotic_initiation_factor_4F eIF4F], which is a ternary complex made up of a cap-binding protein [https://en.wikipedia.org/wiki/EIF4E (eIF4E)] and RNA helicase [https://en.wikipedia.org/wiki/EIF4A (eIF4A)] connected by the bridging protein (eIF4G) (Figure 3). Translation initiation is stimulated by the PABP bound to the poly(A) tail and its association with eIF4G.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> The 5’ UTR is unwound by the elF4F complex, and ribosomes are recruited to create the initiation complex. The eIF4G protein then guides the 40S subunit to the start codon (AUG), which is followed by the binding 60S ribosomal subunit, creating the 80S initiation complex.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> [[Image:closedlooper.png|300px|left|thumb| "Figure 3:" Closed loop model of the eIF4F complex and PABP creating a loop out of the mRNA ]] The association of the PABP and eIF4G gave rise to the name “closed loop.”<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> Mutations of Arg→Ala and Lys→Ala in human eIF4G and in yeast extracts decrease the rate of translation initiation and destabilize the interactions with PABP, indicating that basic residues are essential to the interaction with PABP.<ref name="Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein">Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print. </ref> |
In more complex eukaryotic organisms, PABP indirectly stimulates translation via [https://en.wikipedia.org/wiki/PAIP1 PAIP-1] (PABP interacting protein). A higher presence of PAIP-1 increases the rate of translation initiation, indicating another way to “close the loop.”¹ | In more complex eukaryotic organisms, PABP indirectly stimulates translation via [https://en.wikipedia.org/wiki/PAIP1 PAIP-1] (PABP interacting protein). A higher presence of PAIP-1 increases the rate of translation initiation, indicating another way to “close the loop.”¹ | ||
Revision as of 01:25, 26 April 2018
Contents |
Discussion of Human Poly(A) Binding Protein
Background
PolyAdenosine Binding Protein is a mRNA binding protein that binds to the 3’ Poly(A) tail on mRNA. The human PABP was discovered in 1973 by the use of a sedimentation profile detailing the RNase digestion differentiated the PABP protein. [1] Attempts to purify the 75 kDa protein then followed, where shortly after, in 1983, then considered “poly(A)-organizing protein,” was determined and purified by molecular weight, ligand-binding affinity, and amounts found in cytoplasmic portions of cell with ability to bind to free poly(A). [2]
PABP is comprised of four RNA recognition motifs (RRMs), which are highly conserved RNA-binding domains.[3] The RRM in PABP is found in over two hundred families of proteins across species, indicating that it is ancient.[3] Through extensive Adenosine recognition by the RRMs of PABP, the protein is involved in three main functions: recognition of the 3’ Poly(A) tail, mRNA stabilization, and eukaryotic translation initiation. However, the contributions of controlling gene expression throughout different families of PABPs is not yet fully understood. PABP families are divided into nuclear and cytoplasmic. [4] PABP1, which is predominantly cytoplasmic, is often referred to as PABP because it is the only form of PABP that has been extensively studied in its role with mRNA translation and stability. [4]
Structure
| |||||||||||
Disease and Medical Relevance
Oculopharyngeal Muscular Dystrophy
Oculopharyngeal muscular dystrophy, or OPMD, is an autosomal dominant late-onset disease. [7] The disease is characterized by the myopathy of the eyelids and the throat with symptoms consisting of eye-drooping and difficulty swallowing. There are two types of OPMD: autosomal dominant and recessive, both originating from the mutation of the PABP nuclear 1 (PABPN1) gene located on the long arm of chromosome 14. [7] This mutation results in an abnormally long polyalanine tract, 11-18 alanines, opposed to the normal 10. [7] Patients with longer PABPN1 expansion (more alanines) are on average diagnosed at an earlier in life than patients with a shorter expansion; therefore, expansion size plays a role in OPMD severity and progression. [8] This mutation results in PABPN1 forming clumps in muscle cells that can’t be degraded. [7] It’s suspected that this is a source of cell death for effected cells, however, it has not been concluded why this mutation only affects certain muscle cells.
Studies on Mutations
Studies conducted on Drosophila are common due to 75% conservation between human and Drosophila genomes. Drosophila only encode one cytoplasmic PABP, and its deletion results in embryonic lethality. [4] Similarly, in Caenorhabditis elegans, which have two cytoplasmic PABPs, display 50-80% embryonic lethality with the introduction of an RNAi to one of these PABPs. [4]
References
- ↑ Blobel, Gunter. “A Protein of Molecular Weight 78,000 Bound to the Polyadenylate Region of Eukaryotic Messenger Rnas.” Proceedings of the National Academy of Sciences of the United States of America, vol. 70, no. 3, 1973, pp. 924–8.
- ↑ Baer, Bradford W. and Kornberg, Roger D. "The Protein Responsible for the Repeating Structure of Cytoplasmic Poly(A)-Ribonucleoprotein." The Journal of Cell Biology, vol. 96, no. 3, Mar. 1983, pp. 717-721. EBSCOhost.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 Deo, Rahul C, et al. “Recognition of Polyadenylate RNA by the Poly(A)-Binding Protein.” Cell 98:6. (1999) 835-845. Print.
- ↑ 4.0 4.1 4.2 4.3 4.4 Gorgoni, Barbra, and Gray, Nicola. “The Roles of Cytoplasmic Poly(A)-Binding Proteins in Regulating Gene Expression: A Developmental Perspective.” Briefings in Functional Genomics and Proteomics, vol. 3, no. 2, 1 Aug. 2004, pp. 125–141., doi:10.1093/bfgp/3.2.125.
- ↑ Kühn, Uwe and Elmar, Wahle. “Structure and Function of Poly(a) Binding Proteins.” Bba - Gene Structure & Expression, vol. 1678, no. 2/3, 2004.
- ↑ Wang, Zuoren and Kiledjian, Megerditch. “The Poly(A)-Binding Protein and an mRNA Stability Protein Jointly Regulate an Endoribonuclease Activity.” Molecular and Cellular Biology 20.17 (2000): 6334–6341. Print.
- ↑ 7.0 7.1 7.2 7.3 “Oculopharyngeal Muscular Dystrophy.” NORD (National Organization for Rare Disorders), rarediseases.org/rare-diseases/oculopharyngeal-muscular-dystrophy/.
- ↑ Richard, Pascale, et al. “Correlation between PABPN1 Genotype and Disease Severity in Oculopharyngeal Muscular Dystrophy.” Neurology, vol. 88, no. 4, 2016, pp. 359–365., doi:10.1212/wnl.0000000000003554.