This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1481
From Proteopedia
| Line 25: | Line 25: | ||
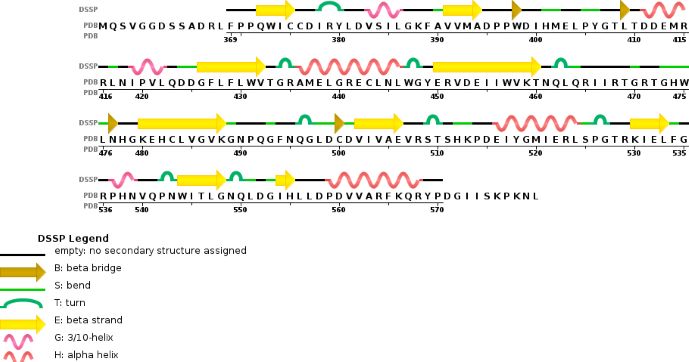
Chain A METTL3'secondary structure, is mainly composed of 20% helical (7 helices; 46 residues)and 24% beta sheet (13 strands; 55 residues) | Chain A METTL3'secondary structure, is mainly composed of 20% helical (7 helices; 46 residues)and 24% beta sheet (13 strands; 55 residues) | ||
| - | [[Image:chain 1 METTL3.jpg]]<ref> | + | [[Image:chain 1 METTL3.jpg]]<ref>[http://www.rcsb.org/pdb/explore/remediatedSequence.do?structureId=5K7M]</ref> |
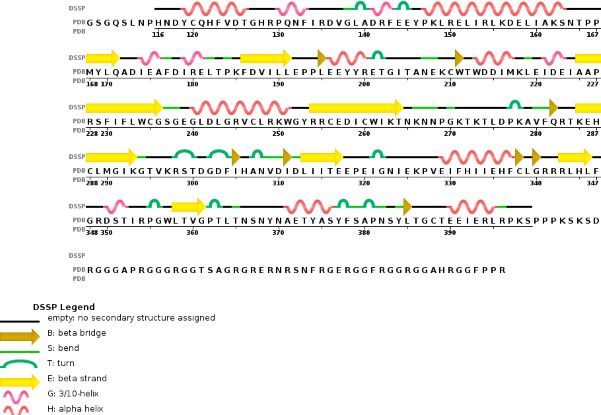
Chain B METTL14 is made of 24% helical s (14 helices; 87 residues) and 18% beta sheet (16 strands; 63 residues) | Chain B METTL14 is made of 24% helical s (14 helices; 87 residues) and 18% beta sheet (16 strands; 63 residues) | ||
| Line 106: | Line 106: | ||
<references/>[https://www.ncbi.nlm.nih.gov/pubmed/27627798] | <references/>[https://www.ncbi.nlm.nih.gov/pubmed/27627798] | ||
<references/>[https://www.ncbi.nlm.nih.gov/pubmed/28121234] | <references/>[https://www.ncbi.nlm.nih.gov/pubmed/28121234] | ||
| - | <references/ | + | <references/> |
<references/> | <references/> | ||
Revision as of 14:06, 29 December 2018
| This Sandbox is Reserved from 06/12/2018, through 30/06/2019 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1480 through Sandbox Reserved 1543. |
To get started:
More help: Help:Editing |
Crystal structure of the catalytic domains of Mettl3/Mettl14 complexInsert caption here
Drag the structure with the mouse to rotate
Insert caption here |
| Drag the structure with the mouse to rotate |
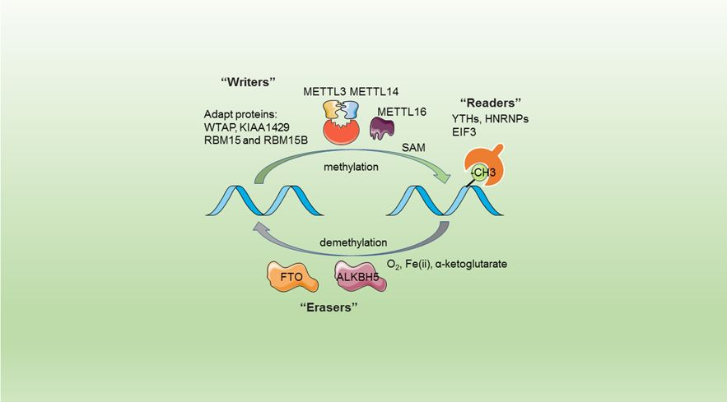
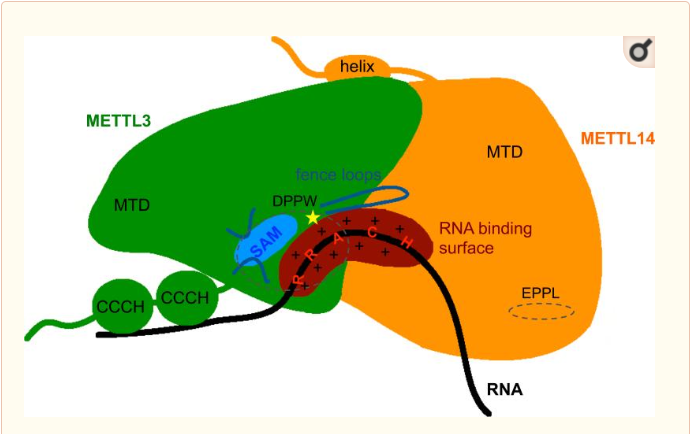
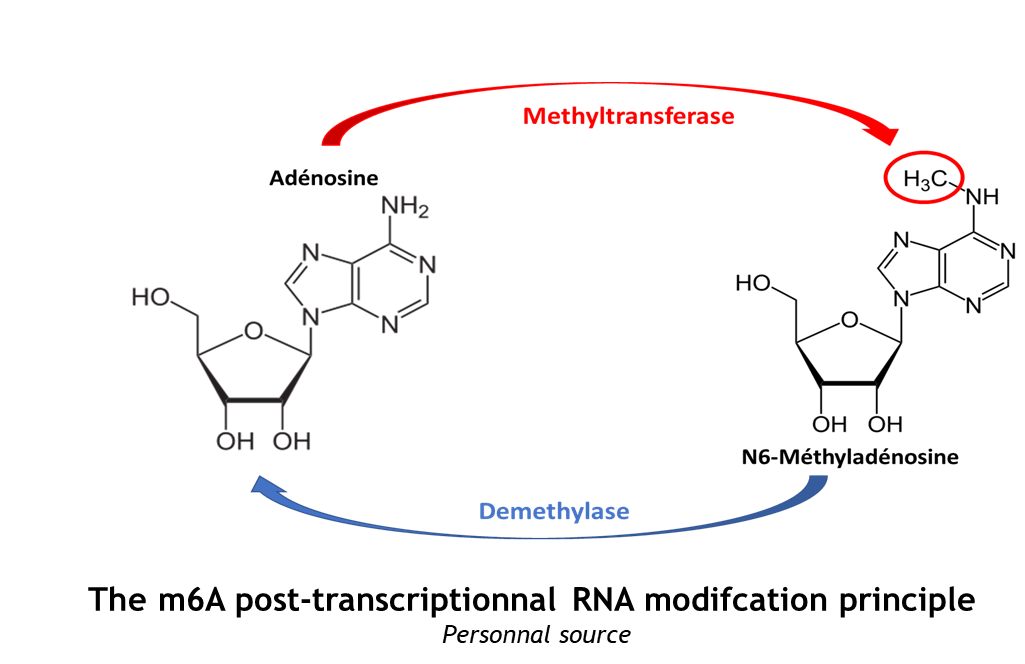
The complex METTL3/METTL14 is a heterodimer enzymatic complex involved into RNA post-transcription modifications by humans. This complex is abble to add a methyl group on adenosin of the RNA, by catalyzing a m6(A) modification.The N(6)-methyladenosine (m(6)A) is a quite common, reversible chemical modifications of RNAs molecules which plays a key role in several biological fonctions. This post transcriptional modification can be added by WRITERS, recognized by READERS and also removed byr ERASERS. The METTL3/METTL14 complex plays the role of writer.
| |||||||||||
References
<Structural Basis for Cooperative Function of Mettl3 and Mettl14 Methyltransferases/>[2] <Structural basis of N(6)-adenosine methylation by the METTL3-METTL14 complex./>[3]
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644
- ↑ [1]