This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1493
From Proteopedia
| Line 41: | Line 41: | ||
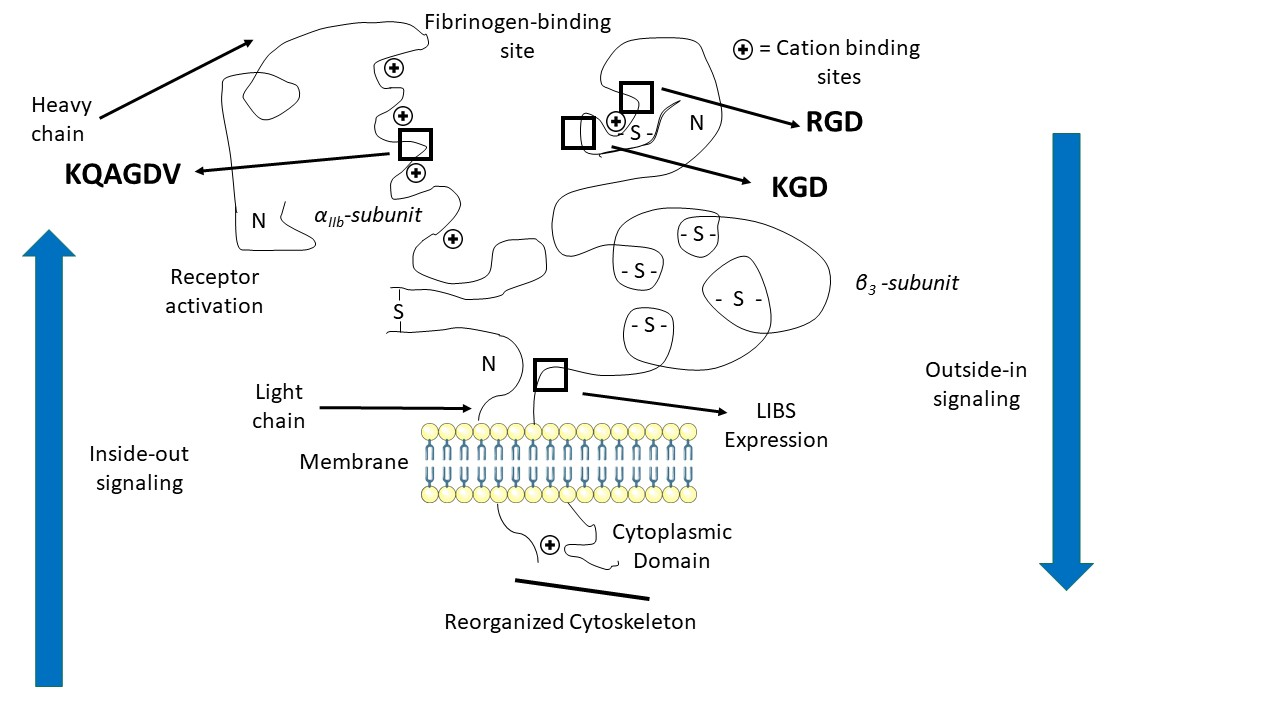
The cytoplasmic tail of the β3 subunit has a NPLY domain which bind proteins with phosphotyrosine binding (PTB) domains. | The cytoplasmic tail of the β3 subunit has a NPLY domain which bind proteins with phosphotyrosine binding (PTB) domains. | ||
| + | [[Image:Binding_sites.png|right|Domains of integrin αIIbβ3]] | ||
== Function == | == Function == | ||
| Line 84: | Line 85: | ||
==== Propagation of activation ==== | ==== Propagation of activation ==== | ||
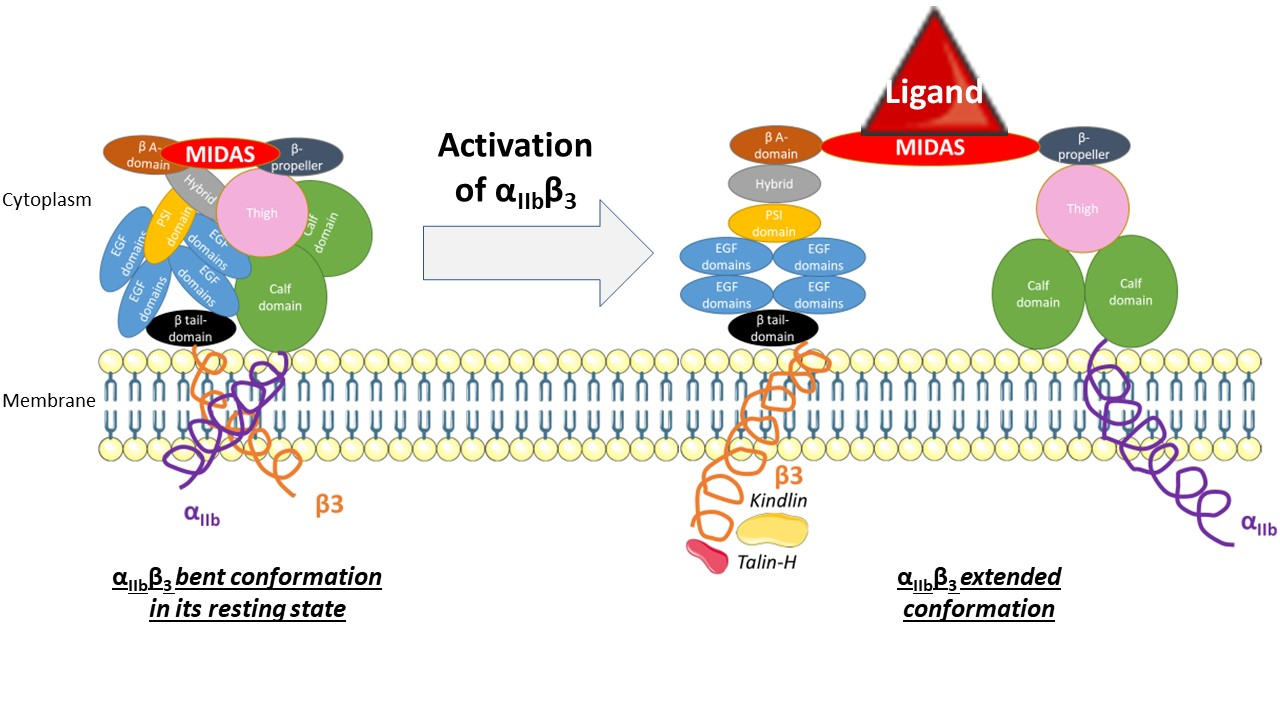
| - | It opens a hinge in the integrin which triggers a very quick succession of subunit shifts that are transmitted from the tail to the extracellular headpiece across the transmembrane domain (inside-out signaling). Movements of helices | + | It opens a hinge in the integrin which triggers a very quick succession of subunit shifts that are transmitted from the tail to the extracellular headpiece across the transmembrane domain (inside-out signaling). Movements of helices and loops moves the headpiece to an extended conformation which uncovers the interface between the two subunits countaining ligand binding sites. Integrin is at an intermediate affinity state and can bind ligands. |
| + | |||
| + | [[Image:Activation.png|right|Activation of the binding site at intermediate affinity]] | ||
The precise mechanisms of activation which occurs in the extracellular part of the integrin remain a mystery. Still, the conformational difference of a disulfide-bonded knot localised in the cysteine-rich core of the β3 subunit between inactivation and activation suggests that this region plays a part in activation. It is thought that the cysteine core of the β3 subunit linked to its N-terminal extremity apply a conformational constraint on the ligand binding site. It includes a few cysteines that remain unpaired and which redox state influence the activation of the integrin, which supposed this region could host a redox site. | The precise mechanisms of activation which occurs in the extracellular part of the integrin remain a mystery. Still, the conformational difference of a disulfide-bonded knot localised in the cysteine-rich core of the β3 subunit between inactivation and activation suggests that this region plays a part in activation. It is thought that the cysteine core of the β3 subunit linked to its N-terminal extremity apply a conformational constraint on the ligand binding site. It includes a few cysteines that remain unpaired and which redox state influence the activation of the integrin, which supposed this region could host a redox site. | ||
==== Activated headpiece ==== | ==== Activated headpiece ==== | ||
| - | Once the ligand binding site has been activated, ligand binding can gradually occur. | + | Once the ligand binding site has been activated, ligand binding can gradually occur. A swing out of the hybrid domain of the β3 subunit around the active site moves the β3 transmembrane domain away from the αIIb transmembrane domain. As a result, the integrin is switched to an open conformation which facilitates binding to ligands. The active site is at its higher affinity state. |
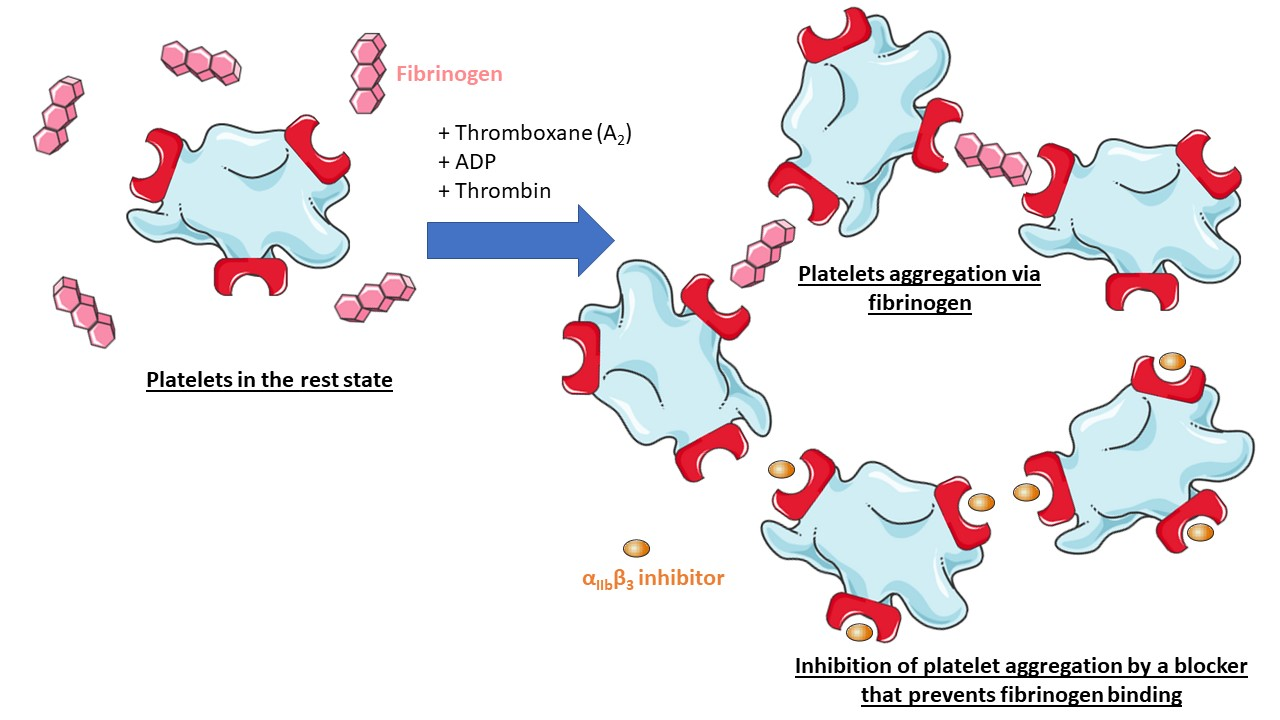
| - | + | The initial contact with the ligand is reversible, but then irreversible binding prevents ligand from dissociating. Binding also changes the conformation of the ligand and may unmask new binding regions on it. | |
| + | Extracellular proteins such as fibrinogen enables platelets aggregation and clotting. Integrin αIIbβ3 also bridges to other αIIbβ3 of adjacent platelets. | ||
| + | [[Image:Clotting.png|right|Clotting]] | ||
Activation mostly occurs via modulation of affinity, but is also affected by avidity for ligand due to receptor clustering by multivalent ligands and changes in membrane fluidity. | Activation mostly occurs via modulation of affinity, but is also affected by avidity for ligand due to receptor clustering by multivalent ligands and changes in membrane fluidity. | ||
Revision as of 00:32, 11 January 2019
| This Sandbox is Reserved from 06/12/2018, through 30/06/2019 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1480 through Sandbox Reserved 1543. |
To get started:
More help: Help:Editing |
Integrin αIIbβ3 (2VDL)
Integrin αIIbβ3 (or glycoprotein IIb/IIIa) is a complex present on the membrane of platelets that intervenes in the activation, adherence and aggregation of platelets during clotting. It is a cation-dependant heterodimeric transmembrane receptor containing a large extracellular headpiece and short intracellular tails. It is synthesized in megakaryocytes.
Its particular shape and localisation on the membrane allows both ligand binding and transduction of the activation signal. It is the dominant integrin on platelets with 70,000 to 90,000 receptors expressed on each platelet in the resting state.
The headpiece (2VDL) of integrin αIIbβ3 enables cation-facilitated ligand binding with multiple ligands (most known being fibrinogen, fibronectin, von Willebrand factors, thrombospondin and vitronectin). Binding affinity is dynamic and depends on the conformational status of the receptor.
| |||||||||||
References
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644